Synthesis of Stabilized Manganese-Containing Nanoparticles with Bombyx mori Chitosan Macromolecules under In Situ Conditions
DOI:
https://doi.org/10.31489/2959-0663/1-26-1Keywords:
manganese nanoparticles, chitosan, Bombyx mori, nanocomposites, in situ synthesis, chemisorption, biopolymers, nanoparticle stabilization, particle morphology, size controlAbstract
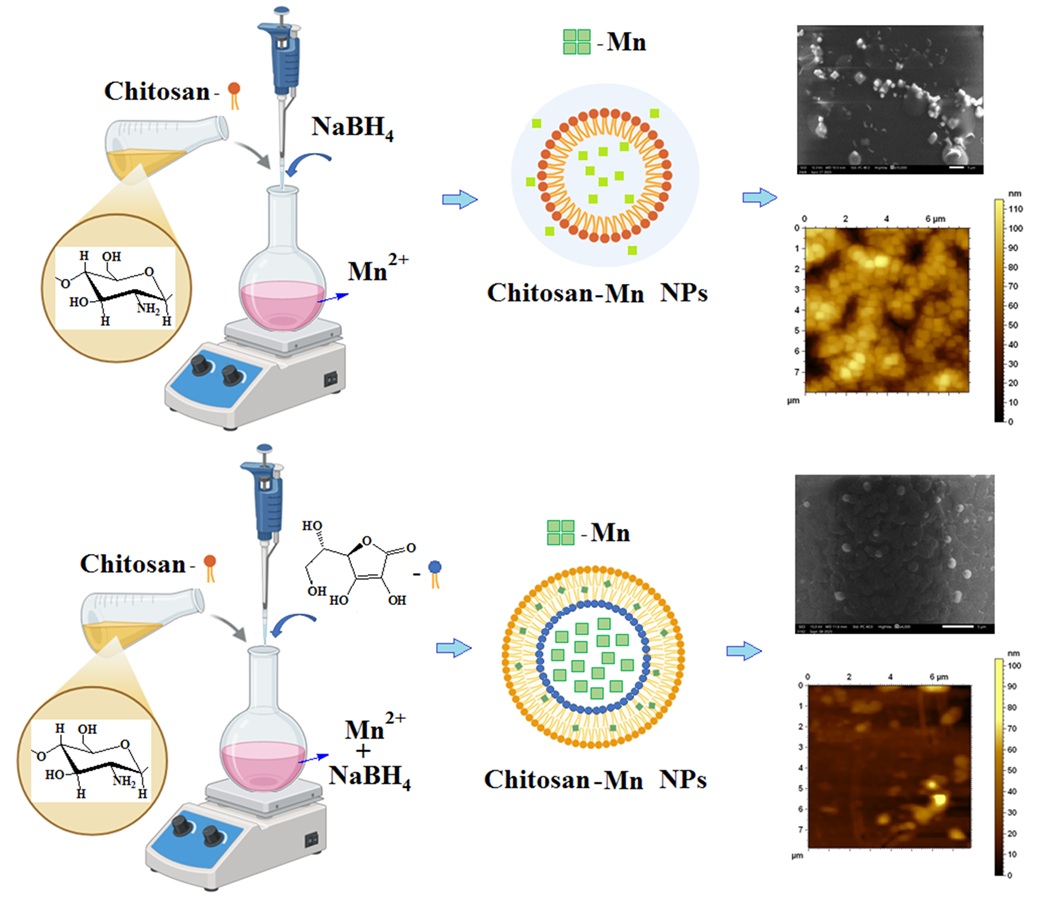
Manganese nanoparticles are highly reactive and tend to aggregate and oxidize, which limits their practical application. Therefore, the aim of this study was to synthesize and stabilize Mn nanoparticles using Bombyx mori chitosan as a natural biopolymer matrix under controlled in situ conditions and to investigate their structural and physicochemical properties. Samples containing Mn nanoparticles stabilized with chitosan in the presence of reducing agents, with hydrodynamic dimensions of 118 nm (97 %) and 144 nm (96 %), were synthesized under in situ conditions. The structural and morphological characteristics were studied using IR spectroscopy, X-ray diffraction (XRD), dynamic light scattering (DLS), and electron microscopy. IR spectroscopic studies revealed local rearrangements within the repeating units of the chitosan polymer chain and interactions between the –NH and –C=O functional groups and manganese-containing nanoparticles. XRD analysis confirmed the reduction of Mn2+ ions. Morphological studies of chitosan–manganese films showed the formation of 50 nm tetragonal and 90 nm spherical metal nanoparticles under the selected synthesis conditions. The results confirm the stabilization of Mn nanoparticles with Bombyx mori chitosan by the chemisorption method and demonstrate effective control of particle size and morphology. Further research should focus on evaluating long-term stability, functional properties, and potential biomedical applications of the synthesized nanocomposites.
References
Tunc, O., Thompson, J., & Tremellen, K. (2010). Development of the NBT assay as a marker of sperm oxidative stress. International Journal of Andrology, 33(1), 13–21. https://doi.org/10.1111/j.1365-2605.2008.00941.x DOI: https://doi.org/10.1111/j.1365-2605.2008.00941.x
Horning, K. J., Caito, S. W., Tipps, K. G., Bowman, A. B., & Aschner, M. (2015). Manganese is essential for neuronal health. Annual Review of Nutrition, 35, 71–108. https://doi.org/10.1146/annurev-nutr-071714-034419 DOI: https://doi.org/10.1146/annurev-nutr-071714-034419
Dorman, D.C., Struve, M.F., James, R.A., McManus, B.E., Marshall, M.W., & Wong, B.A. (2001). Influence of dietary manganese on the pharmacokinetics of inhaled manganese sulfate in male CD rats. Toxicological Sciences, 60(2), 242–251. https://doi.org/10.1093/toxsci/60.2.242 DOI: https://doi.org/10.1093/toxsci/60.2.242
Grujicic, J., & Allen, A.R. (2025). Manganese superoxide dismutase: Structure, function, and implications in human disease. Antioxidants, 14(7), 848. https://doi.org/10.3390/antiox14070848 DOI: https://doi.org/10.3390/antiox14070848
Studer, J. M., Schweer, W. P., Gabler, N. K., & Ross, J. W. (2022). Functions of manganese in reproduction. Animal Reproduction Science, 238, Article 106924. https://doi.org/10.1016/j.anireprosci.2022.106924 DOI: https://doi.org/10.1016/j.anireprosci.2022.106924
Cheema, R.S., Bansal, A.K., Bilaspuri, G.S. (2009). Manganese provides antioxidant protection for sperm cryopreservation that may improve fertilizing ability of buffalo (Bubalus bubalis) spermatozoa. Oxidative Medicine and Cellular Longevity, 2(3), 152–159. https://doi.org/10.4161/oxim.2.3.8804 DOI: https://doi.org/10.4161/oxim.2.3.8804
De Pascali, F., Tréfier, A., Landomiel, F., Bozon, V., Bruneau, G., Yvinec, R., Poupon, A., Crépieux, P., & Reiter, E. (2018). Follicle-stimulating hormone receptor: Advances and remaining challenges. International Review of Cell and Molecular Biology, 338, 1–58. https://doi.org/10.1016/bs.ircmb.2018.02.001 DOI: https://doi.org/10.1016/bs.ircmb.2018.02.001
Lee, B., Pine, M., Johnson, L., Rettori, V., Hiney, J. K., & Dees, W.L. (2006). Manganese acts centrally to activate reproductive hormone secretion and pubertal development in male rats. Reproductive Toxicology, 22(4), 580–585. https://doi.org/10.1016/j.reprotox.2006.03.011 DOI: https://doi.org/10.1016/j.reprotox.2006.03.011
Yang, H., Wang, J., Yang, X., Wu, F., Qi, Z., Xu, B., Liu, W., & Deng, Y. (2019). Occupational manganese exposure, reproductive hormones, and semen quality in male workers: A cross-sectional study. Toxicology and Industrial Health, 35(1), 53–62. https://doi.org/10.1177/0748233718810109 DOI: https://doi.org/10.1177/0748233718810109
Kim, E. A., Cheong, H. -K., Joo, K.-D., Shin, J.-H., Lee, J. S., Choi, S.-B., Kim, M.-O., Lee, I. J., & Kang, D. M. (2007). Effect of manganese exposure on the neuroendocrine system in welders. NeuroToxicology, 28(2), 263–269. https://doi.org/10.1016/j.neuro.2006.07.013 DOI: https://doi.org/10.1016/j.neuro.2006.07.013
Roy, T., Boateng, S. T., Uddin, M. B., Banang-Mbeumi, S., Yadav, R. K., Bock, C. R., Folahan, J. T., Siwe-Noundou, X., Walker, A. L., King, J. A., Buerger, C., Huang, S., & Chamcheu, J. C. (2023).The PI3K-Akt-mTOR and associated signaling pathways as molecular drivers of immune-mediated inflammatory skin diseases: Update on therapeutic strategy using natural and synthetic compounds. Cells, 12(12), 1671. https://doi.org/10.3390/cells12121671 DOI: https://doi.org/10.3390/cells12121671
Srivastava, V.K., Hiney, J. K., Dees, W.L. (2013). Early life manganese exposure upregulates tumor-associated genes in the hypothalamus of female rats: relationship to manganese-induced precocious puberty. Toxicological Sciences, 136(2), 373–381. https://doi.org/10.1093/toxsci/kft195 DOI: https://doi.org/10.1093/toxsci/kft195
Tinkov, A. A., Paoliello, M. M. B., Mazilina, A. N., Skalny, A. V., Martins, A. C., Voskresenskaya, O. N., Aaseth, J., Santamaria, A., Notova, S. V., Tsatsakis, A., Lee, E., Bowman, A. B., & Aschner, M. (2021). Molecular targets of manganese-induced neurotoxicity: A five-year update. International Journal of Molecular Sciences, 22(9), 4646. https://doi.org/10.3390/ijms22094646 DOI: https://doi.org/10.3390/ijms22094646
Peng, Y., & He, Q. (2024). Reproductive toxicity and related mechanisms of micro(nano)plastics in terrestrial mammals: Review of current evidence. Ecotoxicology and Environmental Safety, 279, 116505. https://doi.org/10.1016/j.ecoenv.2024.116505. DOI: https://doi.org/10.1016/j.ecoenv.2024.116505
da Silva, J. D. O., dos Santos, H. C., Bento, G. S., Oliveira, J. F. R., Abud, A. K. S., & Gimenez, I. de F. (2024). Green synthesis of manganese dioxide (MnO2) nanoparticles produced with acerola (Malpighia emarginata) leaf extract. Materials Chemistry and Physics, 315, 128963. https://doi.org/10.1016/j.matchemphys.2024.128963 DOI: https://doi.org/10.1016/j.matchemphys.2024.128963
Tazim, T. Q., Kawsar, M., Hossain, M. S., Bahadur, N. M., & Ahmed, S. (2025). Hydrothermal synthesis of nano-metal oxides for structural modification: A review. Next Nanotechnology, 7, 100167. https://doi.org/10.1016/j.nxnano.2025.100167 DOI: https://doi.org/10.1016/j.nxnano.2025.100167
Huo, Y., Xiu, S., Meng, L.-Y., & Quan, B. (2023). Solvothermal synthesis and applications of micro/nano carbons: A review. Chemical Engineering Journal, 451, 138572. https://doi.org/10.1016/j.cej.2022.138572 DOI: https://doi.org/10.1016/j.cej.2022.138572
Kirubakaran, D., Wahid, J. B. A., Karmegam, N., Jeevika, R., Sellapillai, L., Rajkumar, M., & SenthilKumar, K.J. (2025). A comprehensive review on the green synthesis of nanoparticles: Advancements in biomedical and environmental applications. Biomedical Materials & Devices, 4, 388–413. https://doi.org/10.1007/s44174-025-00295-4 DOI: https://doi.org/10.1007/s44174-025-00295-4
Chaschin, I. S., Perepelkin, E. I., Levin, E. E., Abramchuk, S. S., Anuchina, N. M., Kizas, O. A., Ryzhova, Y. V., & Bakuleva, N. P. (2025). Green synthesis of silver nanoparticles using chitosan in carbonic acid solutions: Effect of pressure and temperature on the structure and antimicrobial properties. Journal of Inorganic and Organometallic Polymers and Materials, 35, 2287–2299. https://doi.org/10.1007/s10904-024-03212-2 DOI: https://doi.org/10.1007/s10904-024-03212-2
Kustov, L., & Vikanova, K. (2023). Synthesis of metal nanoparticles under microwave irradiation: Get much with less energy. Metals, 13(10), 1714. https://doi.org/10.3390/met13101714 DOI: https://doi.org/10.3390/met13101714
Karadi, I., Hiremath, V. J., & Timmanagoudar, S. (2025). Nano ferrites: Synthesis, properties and emerging applications — A comprehensive review. Journal of Advancements in Material Engineering, 10(2), 1–21. https://doi.org/10.46610/JoAME.2025.v10i02.001 DOI: https://doi.org/10.46610/JoAME.2025.v10i02.001
Zhang, X., Sathiyaseelan, A., Naveen, K.V., Lu, Y., & Wang, M.-H. (2023). Research progress in green synthesis of manganese and manganese oxide nanoparticles in biomedical and environmental applications — A review. Chemosphere, 337, 139312. https://doi.org/10.1016/j.chemosphere.2023.139312 DOI: https://doi.org/10.1016/j.chemosphere.2023.139312
Luo, Z.C., Cui, Y.X., Liu, Z.X., Liu, T.L., Yin, F.X., & Zheng, K.H. (2024). Oxidation mechanism of high-manganese heat-resistant steels reinforced by in situ second-phase particles. Corrosion Science, 227, 111720. https://doi.org/10.1016/j.corsci.2023.111720 DOI: https://doi.org/10.1016/j.corsci.2023.111720
Li, K., Li, H., Xiao, T., & Zhang, G. (2020). Zero-valent manganese nanoparticles coupled with different strong oxidants for thallium removal from wastewater. Frontiers of Environmental Science & Engineering, 14(2). https://doi.org/10.1007/s11783-019-1213-5 DOI: https://doi.org/10.1007/s11783-019-1213-5
Akduman, H. Ö., & Özdemir, E. (2025). Zirconia supported bimetallic Co–Mn–B catalyst with superior catalytic activity for hydrolysis of sodium borohydride. International Journal of Hydrogen Energy, 100, 67–78. https://doi.org/10.1016/j.ijhydene.2024.12.261 DOI: https://doi.org/10.1016/j.ijhydene.2024.12.261
Szczyglewska, P., Feliczak-Guzik, A., & Nowak, I. (2023). Nanotechnology–General aspects: A chemical reduction approach to the synthesis of nanoparticles. Molecules, 28(13), 4932. https://doi.org/10.3390/molecules28134932 DOI: https://doi.org/10.3390/molecules28134932
Ullah, R., & Dutta, J. (2008). Photocatalytic degradation of organic dyes with manganese-doped ZnO nanoparticles. Journal of Hazardous Materials, 156(1–3), 194–200. https://doi.org/10.1016/j.jhazmat.2007.12.033 DOI: https://doi.org/10.1016/j.jhazmat.2007.12.033
Bao, C., Serrano-Lotina, A., Niu, M., Portela, R., Li, Y., Lim, K. H., Liu, P., Wang, W. -j., Bañares, M. A., & Wang, Q. (2023). Microwave-associated chemistry in environmental catalysis for air pollution remediation: A review. Chemical Engineering Journal, 466, 142902. https://doi.org/10.1016/j.cej.2023.142902 DOI: https://doi.org/10.1016/j.cej.2023.142902
Zenner, J., Tran, K., Kang, L., Kinzel, N. W., Werlé, C., DeBeer, S., Bordet, A., & Leitner, W. (2024). Synthesis, Characterization, and Catalytic Application of Colloidal and Supported Manganese Nanoparticles. Chemistry — A European Journal, 30, e202304228. https://doi.org/10.1002/chem.202304228 DOI: https://doi.org/10.1002/chem.202304228
Trofimova, O.Y., Ershova, I.V., Maleeva, A.V., et al. (2024). Synthesis and Properties of Manganese(II) and Nickel(II) 1-D Coordination Polymers Based on 2,5-di-hydroxy-3,6-di-tert-butyl-para-quinone. Journal of Inorganic and Organometallic Polymers and Materials, 34, 2779–2787. https://doi.org/10.1007/s10904-024-03013-7 DOI: https://doi.org/10.1007/s10904-024-03013-7
Xu, Z., Zhao, W., Liu, J., Fan, J. (2023). Research on the Surfactant-Assisted Synthesis of MnZn Ferrite Precursor Powders. Magnetochemistry, 9(6), 146. https://doi.org/10.3390/magnetochemistry9060146 DOI: https://doi.org/10.3390/magnetochemistry9060146
Wang, W., Ding, Z., Zhao, X., Wu, S., Li, F., Yue, M., & Liu, J. P. (2015). Microstructure and magnetic properties of MFe2O4 (M = Co, Ni, and Mn) ferrite nanocrystals prepared using colloid mill and hydrothermal method. Journal of Applied Physics, 117(17), 17A328. https://doi.org/10.1063/1.4917463 DOI: https://doi.org/10.1063/1.4917463
Hu, M., Yan, X., Hu, X., Feng, R., & Zhou, M. (2019). Synthesis of silver decorated silica nanoparticles with rough surfaces as adsorbent and catalyst for methylene blue removal. Journal of Sol-Gel Science and Technology, 89(3), https://doi.org/10.1007/s10971-018-4871-z DOI: https://doi.org/10.1007/s10971-018-4871-z
Rostami, S., Mehdinia, A., & Jabbari, A. (2017). Seed-mediated grown silver nanoparticles as a colorimetric sensor for detection of ascorbic acid. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 180, 204–210. https://doi.org/10.1016/j.saa.2017.03.020 DOI: https://doi.org/10.1016/j.saa.2017.03.020
Varin, R. A.; Mattar, D. K.; Bidabadi, A. S.; Polański, M. K. (2017). Synthesis of amorphous manganese borohydride in the (NaBH4–MnCl2) system, its hydrogen generation properties and crystalline transformation during solvent extraction. Journal of Energy Chemistry, 26(1), 24–34. http://dx.doi.org/10.1016/j.jechem.2016.08.011 DOI: https://doi.org/10.1016/j.jechem.2016.08.011
Wang, M.; Pang, P.; Koopal, L. K.; Suib, S.; Wang, Y.; Liu, F. (2014). One-step synthesis of δ‑MnO2 nanoparticles using ascorbic acid and their scavenging properties to Pb(II), Zn(II) and methylene blue. Materials Chemistry and Physics, 148(3), 1149–1156. https://doi.org/10.1016/j.matchemphys.2014.09.037 DOI: https://doi.org/10.1016/j.matchemphys.2014.09.037
Siddique, M. A. R., Khan, M. A., Bokhari, S. A. I., Ismail, M., Ahmad, K., Haseeb, H. A., Kayani, M. M., Khan, S., Zahid, N., & Khan, S.B. (2024). Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents. Green Processing and Synthesis, 13, 20230158. https://doi.org/10.1515/gps-2023-0158 DOI: https://doi.org/10.1515/gps-2023-0158
Patra, S. K., Molla, M. R., Singh, A. K., Ghosh, P. K., & Jana, R. N. (2021). Synthesis and purification of metal nanoparticles by membrane filtration. Journal of Inorganic and Organometallic Polymers and Materials, 31(4), 1357–1366. https://doi.org/10.1007/s10904-021-01927-3
Alam, M. A., Ahmed, S., Bishwas, R. K., Mostofa, S., & Jahan, S. A. (2025). X-ray crystallographic diffraction study by whole powder pattern fitting (WPPF) method: Refinement of crystalline nanostructure polymorphs TiO2. South African Journal of Chemical Engineering, 51, 68–77. https://doi.org/10.1016/j.sajce.2024.10.010 DOI: https://doi.org/10.1016/j.sajce.2024.10.010
Ben Amor, I., Hemmami, H., Grara, N., Aidat, O., Ben Amor, A., Zeghoud, S., & Bellucci, S. (2024). Chitosan: A Green Approach to Metallic Nanoparticle/Nanocomposite Synthesis and Applications. Polymers, 16(18), 2662. https://doi.org/10.3390/polym16182662 DOI: https://doi.org/10.3390/polym16182662
Eltaweil, A. S., El-Tawil, A. M., Abd El-Monaem, E. M., & El-Subruiti, G. M. (2021). Zero Valent Iron Nanoparticle-Loaded Nanobentonite Intercalated Carboxymethyl Chitosan for Efficient Removal of Both Anionic and Cationic Dyes. ACS Omega, 6(9), 6348–6360. https://doi.org/10.1021/acsomega.0c06251 DOI: https://doi.org/10.1021/acsomega.0c06251
Rinaudo, M. (2006). Chitin and chitosan: Properties and applications. Progress in Polymer Science, 31(7), 603–632. https://doi.org/10.1016/j.progpolymsci.2006.06.001 DOI: https://doi.org/10.1016/j.progpolymsci.2006.06.001
Sajna, P., Varma, A., & Vasudevan, G. (2008). Application of Spectroscopic Methods for Structural Analysis of Chitin and Chitosan. Marine Drugs, 8(5), 1567–1587. https://doi.org/10.3390/md8051567 DOI: https://doi.org/10.3390/md8051567
Hu, Z., Lu, S., Cheng, Y., Kong, S., Li, S., Li, C., & Yang, L. (2019). Investigation of the effects of molecular parameters on the hemostatic properties of chitosan. International Journal of Biological Macromolecules, 136, 327–336. https://doi.org/10.3390/molecules23123147 DOI: https://doi.org/10.3390/molecules23123147
Wang, Y., Chen, X., Liu, J., Zhang, L., & Zhao, X. (2021). Synthesis of chitosan–metal nanocomposites for biomedical applications. Materials Science and Engineering: C, 124, 112051. https://doi.org/10.1016/j.msec.2021.112051 DOI: https://doi.org/10.1016/j.msec.2021.112051
Filippov, S. K., Khusnutdinov, R., Murmiliuk, A., Inam, W., Zakharova, L. Ya., Zhang, H., & Khutoryanskiy, V. V. (2023). Dynamic light scattering and transmission electron microscopy in drug delivery: A roadmap for correct characterization of nanoparticles and interpretation of results. Materials Horizons, 10, 5354–5370. https://doi.org/10.1039/d3mh00717k DOI: https://doi.org/10.1039/D3MH00717K
Kim, S. et al. (2020). Structural insights into chitosan–ZnO nanocomposites using FTIR and XRD. Journal of Molecular Structure, 1212, 128118. https://doi.org/10.1016/j.molstruc.2020.128118
Faizan, M., Naz, M.Y., Shah, S.A.S.A., Shakir, I., Khaliq, M., Busharat, M.A., Pan, D. (2025). Mesoporous magnetic MnFe2O4@SiO2-chitosan nanocomposite for efficient adsorptive removal of Zn(II) and Cd(II) ions from aqueous media. Desalination and Water Treatment, 324, 101488. https://doi.org/10.1016/j.dwt.2025.101488 DOI: https://doi.org/10.1016/j.dwt.2025.101488
Rodrigues de Sá, M., Mendes da Silva, T., Souza, E.S., Soares de Carvalho, A., Filho, M.R.A.A., Silva-Araújo, E.R. (2025). Magnetic and fluorescent manganese silicate nanostructures for advanced applications. ChemistrySelect, 10(24), e02098. https://doi.org/10.1002/slct.202502098 DOI: https://doi.org/10.1002/slct.202502098
Kumar, S., Yadav, B., Sharma, P., Singh, R. & Gupta, A. (2021). Preparation and Characterization of Chitosan-Coated Manganese Ferrospinel Nanoparticles Conjugated with Laccase for Environmental Bioremediation. Polymers, 13(22), 3921. https://doi.org/10.3390/polym13223921 DOI: https://doi.org/10.3390/polym13091453
Lahouti, S., & Naeimi, H. (2020). Chitosan-encapsulated manganese ferrite particles bearing sulfonic acid group catalyzed efficient synthesis of spiro indenoquinoxalines. RSC Advances, 10(55), 33334–33343. https://doi.org/10.1039/d0ra04925e DOI: https://doi.org/10.1039/D0RA04925E
Nguyen, T. V., Le, H. T., Pham, H. T., & Nguyen, D. T. (2023). Chitosan based nanosorbents for removal of heavy metals: Cd, Co, Cu, and Pb. Scientific Reports, 13, Article 16958. https://doi.org/10.1038/s41598-023-44014-5
Patel, R., Mehta, A. & Joshi, M. (2018). Synthesis of MnFe2O4@chitosan nanocomposites for magnetic hyperthermia and drug delivery applications. Materials, 11(12), 2542. https://doi.org/10.3390/ma11122542 DOI: https://doi.org/10.3390/ma11122542
Zhang, Q., Liu, J., Wang, X., Li, Y. & Chen, H. (2021). Catalytic application of Pd–Chitosan@MnFe2O4 nanocomposites: Structure and interaction analysis. Journal of Molecular Liquids, 339, 117222. https://doi.org/10.1016/j.molliq.2021.117222 DOI: https://doi.org/10.1016/j.molliq.2021.117222
Goy, R. C., de Britto, D., & Assis, O. B. G. (2009). A review of the antimicrobial activity of chitosan. Polímeros, 19(3), 241–247. https://doi.org/10.1590/S0104-14282009000300006 DOI: https://doi.org/10.1590/S0104-14282009000300013
Niu, Y., Hu, W. (2024). Preparation, characterization and application in environmental protection of low-molecular-weight chitosan: a review. Sustain Environ Res 34, 29. https://doi.org/10.1186/s42834-024-00236-8 DOI: https://doi.org/10.1186/s42834-024-00236-8
Podgorbunskikh, E., Kuskov, T., Rychkov, D., Lomovskii, O., & Bychkov, A. (2022). Mechanical amorphization of chitosan with different molecular weights. Polymers, 14(20), 4438. https://doi.org/10.3390/polym14204438 DOI: https://doi.org/10.3390/polym14204438
Lawson, A. C., Vandervoort, K. G., Welp, U., & Hinks, D.G. (1994). Magnetic structure of α-manganese. Journal of Applied Physics, 76(10), 7049–7051. https://doi.org/10.1063/1.358261 DOI: https://doi.org/10.1063/1.358024
Yamauchi, H., Sari, D. P., Watanabe, I., & Yasui, Y. (2020). High-temperature short-range order in Mn3RhSi. Communications Materials, 1, 43. https://doi.org/10.1038/s43246-020-0042-1 DOI: https://doi.org/10.1038/s43246-020-0042-1
Pardeep, Bitla, Y., Lalita, Patra, A. K., & Basheed, G. A. (2023). In-field critical behaviour of β-Mn type Co–Zn–Mn skyrmion-host. Physica B: Condensed Matter, 654, 414669. https://doi.org/10.1016/j.physb.2023.414669 DOI: https://doi.org/10.1016/j.physb.2023.414669
Hornfeck, W., & Kuhn, P. (2014). Octagonal symmetry in low-discrepancy β-manganese. Acta Crystallographica Section A Foundations and Advances, 70(5), 441–447. https://doi.org/10.1107/s2053273314009218 DOI: https://doi.org/10.1107/S2053273314009218
Karube, K., White, J. S., Ukleev, V., Dewhurst, C. D., Cubitt, R., Kikkawa, A., Tokunaga, Y., Rønnow, H. M., Tokura, Y., & Taguchi, Y. (2020). Metastable skyrmion lattices governed by magnetic disorder and anisotropy in β-Mn-type chiral magnets. Physical Review B, 102(6), 064408. https://doi.org/10.1103/PhysRevB.102.064408 DOI: https://doi.org/10.1103/PhysRevB.102.064408
Bakshi, P. S., Singha, M., & Rathi, P. (2013). Structural modification in chitosan after metal oxide incorporation: An XRD analysis. Carbohydrate Polymers, 98, 599–606. https://doi.org/10.1016/j.carbpol.2013.07.083 DOI: https://doi.org/10.1016/j.carbpol.2013.07.083
Wang, X., Chen, X., Zhang, Y., Li, Y., & Zhao, J. (2015). Synthesis and characterization of Mn nanoparticles via reduction of MnCl2. Journal of Alloys and Compounds, 648, 837–843. https://doi.org/10.1016/j.jallcom.2015.07.164 DOI: https://doi.org/10.1016/j.jallcom.2015.07.164
Rasaee, M. J., Esfandiari, M., Soleimani, M., Azimi, S., & Fazeli, M. (2022). Structural and antimicrobial properties of chitosan‑based nanoparticles. Polymers, 14(24), 5324. https://doi.org/10.3390/polym14245324
Shoemaker, C. B., Shoemaker, D. P., Hopkins, T. E., & Yindepit, S. (1978). Refinement of the structure of β-manganese and of a related phase in the Mn–Ni–Si system. Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry, 34(12), 3573–3576. https://doi.org/10.1107/s0567740878011620 DOI: https://doi.org/10.1107/S0567740878011620
Pandey, R., & Shukla, S. (2023). Nanocomposites of chitosan with transition metals: synthesis and physicochemical properties. Materials Chemistry and Physics, 303, 127596. https://doi.org/10.1016/j.matchemphys.2023.127596 DOI: https://doi.org/10.1016/j.matchemphys.2023.127596
Liu, Y., Zhang, X., Li, J., & Wang, S. (2021). Structural properties of chitosan‑based nanocomposites with transition metals. Polymers, 13(16), 2714. https://doi.org/10.3390/polym13162714 DOI: https://doi.org/10.3390/polym13162714
Zhuo, Shujuan; Fang, Jing; Li, Meng; Wang, Jing; Zhu, Changqing; Du, Jinyan (2019). Manganese(II)-doped carbon dots as effective oxidase mimics for sensitive colorimetric determination of ascorbic acid. Microchimica Acta, 186 (745). https://doi.org/10.1007/s00604 019 3887 6 DOI: https://doi.org/10.1007/s00604-019-3887-6
Deshmukh, Aarti R.; Kim, Beom Soo. (2019). Chitosan–Vitamin C Nanoparticles. KSBB Journal, 34 (4), 221–232. https://doi.org/10.7841/ksbbj.2019.34.4.221 DOI: https://doi.org/10.7841/ksbbj.2019.34.4.221
Podgorbunskikh, E., et al. (2022). Mechanical Amorphization of Chitosan with Different Molecular Weights. Polymers, 14(20), 4438. https://doi.org/10.3390/polym14204438 DOI: https://doi.org/10.3390/polym14204438
Nosal, W.H., Thompson, D.W., Yan, L., Sarkar, S., Subramanian, A., Woollam, J.A. (2005). Infrared optical properties and AFM of spin-cast chitosan films chemically modified with 1,2 epoxy-3-phenoxy-propane. Colloids and Surfaces B: Biointerfaces, 46(1), 26–31. https://doi.org/10.1016/j.colsurfb.2005.08.006 DOI: https://doi.org/10.1016/j.colsurfb.2005.08.006
Ferreira, A. M., et al. (2023). AFM surface characterization of chitosan films for biomedical applications. Nanomaterials, 13(4), 720. https://doi.org/10.3390/nano13040720 DOI: https://doi.org/10.3390/nano13040720
Li, Z., Gao, K., Han, G., Wang, R., Li, H., Zhaoabc X.S., Guo. P. (2015). Solvothermal synthesis of MnFe2O4 colloidal nanocrystal assemblies and their magnetic and electrocatalytic properties. New Journal of Chemistry, 39, 361–368. https://doi.org/10.1039/C4NJ01466A DOI: https://doi.org/10.1039/C4NJ01466A
Wang, L., Hu, C., Nemoto, Y., Tateyama, Y., & Yamauchi, Y. (2010). On the role of ascorbic acid in the synthesis of single-crystal hyperbranched platinum nanostructures. Crystal Growth & Design, 10(8), 3454–3460. https://doi.org/10.1021/cg100207q DOI: https://doi.org/10.1021/cg100207q
Elshoky, H. A., Salaheldin, T. A., Ali, M. A., & Gaber, M. H. 2018). Ascorbic acid prevents cellular uptake and improves biocompatibility of chitosan nanoparticles. International Journal of Biological Macromolecules, 115, 358–366. (https://doi.org/10.1016/j.ijbiomac.2018.04.055 DOI: https://doi.org/10.1016/j.ijbiomac.2018.04.055
Plikeva, S., Ivanov, D., Petrova, T., & Kolev, D. (2021). Preparation and characterization of chitosan‐coated manganese‐ferrite nanoparticles. Polymers, 13(9), 1453. https://doi.org/10.3390/polymers13091453 DOI: https://doi.org/10.3390/polym13091453
Ebadi, M., Asikin-Mijan, N., Jamil, M.S. Md., Iqbal, A., Yousif, E., Zain, A.R. Md, Aziz, T. H. T., & Rahimi Yusop, M. (2023). Palladium nanoparticles on chitosan coated superparamagnetic manganese ferrite. Polymers, 15(1), 232. https://doi.org/10.3390/polym15010232 DOI: https://doi.org/10.3390/polym15010232
Rashid, K., Ahmed, S., & Khan, M. (2020). Chitosan-encapsulated manganese ferrite bearing sulfonic acid catalyzed spiro indenoquinoxaline synthesis. RSC Advances, 10, 4925. https://doi.org/10.1039/D0RA04925E DOI: https://doi.org/10.1039/D0RA04925E
Kumar, R., Chen, S.-M., & Lee, C.-H. (2016). Carbon nanotubes decorated with manganese nanoparticles for electrochemical determination of vitamin C. Journal of Alloys and Compounds, 648, 837–843. https://doi.org/10.1016/j.jallcom.2015.12.110 DOI: https://doi.org/10.1016/j.jallcom.2015.12.110
Patel, D., Singh, U., & Ghosh, S. (2023). Nanohybrid based on Mn0.8Zn0.2Fe2O4 functionalized with chitosan and sodium alginate for curcumin loading. AAPS PharmSciTech. https://doi.org/10.1208/s12249-023-02683-9 DOI: https://doi.org/10.1208/s12249-023-02683-9
Davidson, E., Pereira, J., Giannelli, G. G., Murphy, Z., Anagnostopoulos, V., & Santra, S. (2023).Multi-functional chitosan nanovesicles loaded with bioactive manganese for potential wound healing applications. Molecules, 28(16), 6098. https://doi.org/10.3390/molecules28166098 DOI: https://doi.org/10.3390/molecules28166098
Turck, D., Bohn, T., Castenmiller, J., de Henauw, S., Hirsch‐Ernst, K., Knutsen, H. K., Maciuk, A., Mangelsdorf, I., McArdle, H. J., Pentieva, K., Siani, A., Thies, F., Tsabouri, S., Vinceti, M., Bornhorst, J., Cubadda, F., Dopter, A., FitzGerald, R., … Naska, A. (2023). Scientific opinion on the tolerable upper intake level for manganese. EFSA Journal, 21(12). https://doi.org/10.2903/j.efsa.2023.8413 DOI: https://doi.org/10.2903/j.efsa.2023.8413
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Kandiyor Kh. Ergashev, Noira R. Vokhidova, Sayyora Sh. Rashidova

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



