Bombyx mori Chitosan–Caffeine Nanocapsules: Formation, Structural Features, and Physicochemical Properties
DOI:
https://doi.org/10.31489/2959-0663/1-26-2Keywords:
chitosan, Bombyx mori, caffeine, nanoparticles, self-assembly, drug delivery systems, core–shell structures, controlled releaseAbstract
This study aimed to engineer Bombyx mori chitosan–caffeine nanocapsules via a self-assembly approach and to comprehensively characterize their structural architecture, physicochemical properties, and release kinetics under precisely controlled pH conditions. Chitosan–caffeine nanocapsules were synthesized in aqueous medium at low temperature under controlled acidic conditions (pH 3.2 and 4.5), achieving yields of 91–94 %. Their physicochemical properties were analyzed using UV and FTIR spectroscopy, X-ray diffraction, scanning electron microscopy, and transmission electron microscopy. The results showed that hydrogen bonding and electrostatic interactions between the protonated amino groups of chitosan and the carbonyl groups of caffeine promoted the formation of spherical and oval nanostructures with diameters in the range of
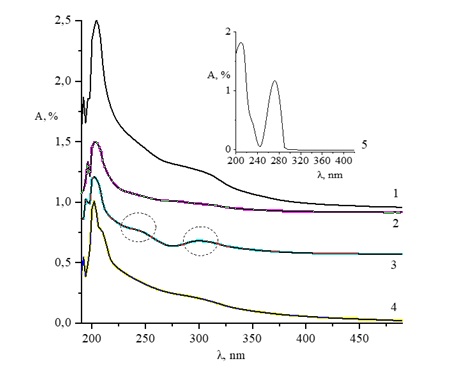
100–400 nm. Spectroscopic analysis confirmed the intermolecular interactions responsible for encapsulation, while X-ray diffraction indicated a decrease in caffeine crystallinity after incorporation into the polymer matrix. SEM and TEM micrographs demonstrated efficient encapsulation of caffeine within the chitosan matrix and confirmed the formation of stable core–shell nanostructures. Release profile analysis showed that approximately 20 % of the total caffeine content was released from the chitosan nanocapsules over the monitored period, confirming their controlled release behavior. These chitosan-caffeine nanocapsules hold potential for future use in the development of drugs with prolonged release properties.
References
Abdoli, F., Davoudi, M., Momeni, F., et al. (2024). Estimate the prevalence of daily caffeine consumption, caffeine use disorder, caffeine withdrawal, and perceived harm in Iran: a cross-sectional study. Scientific Reports, 14, 7644. https://doi.org/10.1038/s41598-024-58496-8 DOI: https://doi.org/10.1038/s41598-024-58496-8
Seyedabadi, M. M., Rostami, H., Jafari, S. M., & Fathi, M. (2021). Development and characterization of chitosan-coated nanoliposomes for encapsulation of caffeine. Food Bioscience, 40, 100857. https://doi.org/10.1016/j.fbio.2020.100857 DOI: https://doi.org/10.1016/j.fbio.2020.100857
Peng, H., Brown, M., Bowdler, P., & Honeychurch, K. C. (2020). Extraction-free, direct determination of caffeine in microliter volumes of beverages by thermal desorption–gas chromatography–mass spectrometry. International Journal of Analytical Chemistry, 2020, 5405184. https://doi.org/10.1155/2020/5405184 DOI: https://doi.org/10.1155/2020/5405184
Chow, C. H., Kan, Y. C., & Ho, K. S. (2019). A simple and rapid gas chromatographic method for routine caffeine determination in beverages using a nitrogen–phosphorus detector. Journal of Analytical Chemistry, 74, 764–770. https://doi.org/10.1134/S1061934819080045 DOI: https://doi.org/10.1134/S1061934819080045
Radeva, L., Kalampalika, E., Yordanov, Y., Petrov, P. D., Tzankova, V., & Yoncheva, K. (2025). Formulation of caffeine–hydroxypropyl-β-cyclodextrin complex in hydrogel for skin treatment. Gels, 11, 326. https://doi.org/10.3390/gels11050326 DOI: https://doi.org/10.3390/gels11050326
Kashapov, R. R., Kashapova, N. E., Ziganshina, A. Y., Syakaev, V. V., Khutoryanskiy, V. V., & Zakharova, L. Y. (2021). Interaction of mucin with viologen and acetate derivatives of calix[4]resorcinols. Colloids and Surfaces B: Biointerfaces, 208, 112089. https://doi.org/10.1016/j.colsurfb.2021.112089 DOI: https://doi.org/10.1016/j.colsurfb.2021.112089
Chen, X.-M., Chen, Y., Hou, X.-F., Wu, X., Gu, B.-H., & Liu, Y. (2018). Sulfonato-β-cyclodextrin mediated supramolecular nanoparticle for controlled release of berberine. ACS Applied Materials & Interfaces, 10, 25555–25563. https://doi.org/10.1021/acsami.8b08651 DOI: https://doi.org/10.1021/acsami.8b08651
Schneider, H.-J. (2009). Binding mechanisms in supramolecular complexes. Angewandte Chemie International Edition, 48(22), 3924–3977. https://doi.org/10.1002/anie.200802947 DOI: https://doi.org/10.1002/anie.200802947
Ibrahim, A. G., Elgammal, W. E., & Hassan, S. M. (2024). Development of a chitosan derivative bearing the thiadiazole moiety and evaluation of its antifungal and larvicidal efficacy. Polymer Bulletin, 81(2), 1291–1302. https://doi.org/10.1007/s00289-023-04765-x DOI: https://doi.org/10.1007/s00289-023-04765-x
Naskar, S., Sharma, S., & Kuotsu, K. (2019). Chitosan-based nanoparticles: An overview of biomedical applications and their preparation. Journal of Drug Delivery Science and Technology, 49, 66–81. https://doi.org/10.1016/j.jddst.2018.10.022 DOI: https://doi.org/10.1016/j.jddst.2018.10.022
Duan, C., Meng, X., Meng, J., et al. (2019). Chitosan as a preservative for fruits and vegetables: a review on chemistry and antimicrobial properties. Journal of Bioresources and Bioproducts, 4, 11–21. https://doi.org/10.21967/jbb.v4i1.189 DOI: https://doi.org/10.21967/jbb.v4i1.189
Fouda, M. M., Wittke, R., Knittel, D., & Schollmeyer, E. (2009). Use of chitosan/polyamine biopolymers based on cotton as a model system to prepare antimicrobial wound dressing. International Journal of Diabetes Mellitus, 1, 61–64. https://doi.org/10.1016/j.ijdm.2009.05.005 DOI: https://doi.org/10.1016/j.ijdm.2009.05.005
Shaban, N. Z., Aboelsaad, A. M., Shoueir, K. R., et al. (2020). Chitosan-based dithiophenolato nanoparticles: preparation, mechanistic information of DNA binding, antibacterial, and cytotoxic activities. Journal of Molecular Liquids, 318, 114252. https://doi.org/10.1016/j.molliq.2020.114252 DOI: https://doi.org/10.1016/j.molliq.2020.114252
Kou, W., Yang, Y., Fan, H., et al. (2024). Regeneration of dental pulp via collagen hydrogel composited with resveratrol-loaded chitosan nanoparticle in a rabbit model of dental pulp injury. Polymer Bulletin, 81, 14235–14248. https://doi.org/10.1007/s00289-024-05318-6 DOI: https://doi.org/10.1007/s00289-024-05318-6
Zakharova, N. V., Simonova, M. A., Zelinskii, S. N., et al. (2019). Synthesis, molecular characteristics, and stimulus-sensitivity of graft copolymer of chitosan and poly(N,N-diethylacrylamide). Journal of Molecular Liquids, 292, 111355. https://doi.org/10.1016/j.molliq.2019.111355 DOI: https://doi.org/10.1016/j.molliq.2019.111355
Hu, T., Ma, Y., Huang, J., et al. (2020). Self-organized thermo-responsive poly(lactic-co-glycolic acid)-graft-pullulan nanoparticles for synergistic thermo-chemotherapy of tumor. Carbohydrate Polymers, 237, 116104. https://doi.org/10.1016/j.carbpol.2020.116104 DOI: https://doi.org/10.1016/j.carbpol.2020.116104
Roy, H., Nayak, B. S., & Nandy, S. (2020). Chitosan-anchored nanoparticles in current drug development utilizing computer-aided pharmacokinetic modeling: Case studies for target-specific cancer treatment and future perspectives. Current Pharmaceutical Design, 26, 1666–1675. https://doi.org/10.2174/1381612826666200203121241 DOI: https://doi.org/10.2174/1381612826666200203121241
Khan, M. M., Madni, A., Torchilin, W., et al. (2019). Lipid–chitosan hybrid nanoparticles for controlled delivery of cisplatin. Drug Delivery, 26, 765–772. https://doi.org/10.1080/10717544.2019.1642420 DOI: https://doi.org/10.1080/10717544.2019.1642420
Francescato, G., Leitão, M. I. P. S., Orsini, G., & Petronilho, A. (2024). Synthesis and medicinal applications of N heterocyclic carbene complexes based on caffeine and others. ChemMedChem, 19, e202400118. https://doi.org/10.1002/cmdc.202400118 DOI: https://doi.org/10.1002/cmdc.202400118
Lessa, E. F., Nunes, M. L., & Fajardo, A. R. (2018). Chitosan/waste coffee-grounds composite: An efficient and eco-friendly adsorbent for removal of pharmaceutical contaminants from water. Carbohydrate Polymers, 189, 257–266. https://doi.org/10.1016/j.carbpol.2018.02.018 DOI: https://doi.org/10.1016/j.carbpol.2018.02.018
Mehrabi-Khozani, Z., Jafari, S. M., Sarabandi, K., Rezaei, A., & Maghsoudlou, Y. (2024). Stabilization of caffeine-loaded nanoliposomes via chitosan coating and spray drying for food product applications. Carbohydrate Polymer Technologies and Applications, 8, 100617. https://doi.org/10.1016/j.carpta.2024.100617 DOI: https://doi.org/10.1016/j.carpta.2024.100617
Xu, Y., Kim, C. S., Saylor, D. M., & Koo, D. (2017). Polymer degradation and drug delivery in PLGA-based drug–polymer applications: A review of experiments and theories. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 105(6), 1692–1716. https://doi.org/10.1002/jbm.b.33648 DOI: https://doi.org/10.1002/jbm.b.33648
Zhang, H., Oh, M., Allen, C., & Kumacheva, E. (2020). Monodisperse chitosan nanoparticles for mucosal drug delivery. Biomacromolecules, 21(6), 2244–2252. https://doi.org/10.1021/bm0496211 DOI: https://doi.org/10.1021/bm0496211
Alvarez-Lorenzo, C., & Concheiro, A. (2014). Smart drug delivery systems: From fundamentals to the clinic. Chemical Communications, 50, 7743–7765. https://doi.org/10.1039/C4CC01429D DOI: https://doi.org/10.1039/C4CC01429D
Song, X., Singh, M., Lee, K. E., Vinayagam, R., & Kang, S. G. (2024). Caffeine: A Multifunctional Efficacious Molecule with Diverse Health Implications and Emerging Delivery Systems. International Journal of Molecular Sciences, 25(22), 12003. https://doi.org/10.3390/ijms252212003 DOI: https://doi.org/10.3390/ijms252212003
Paul, B., Xie, L., Yahia, Z. O., & Chen, W. (2025). Recent Review on the Stability of Bioactive Substances Through Encapsulation and Their Application in Dairy Products. Food Reviews International, 42(2), 605–631. https://doi.org/10.1080/87559129.2025.2492338 DOI: https://doi.org/10.1080/87559129.2025.2492338
Layek, B., & Das, S. (2021). Chitosan-based nanomaterials in drug delivery applications. Biopolymer-Based Nanomaterials in Drug Delivery and Biomedical Applications, 185–219. https://doi.org/10.1016/b978-0-12-820874-8.00001-4 DOI: https://doi.org/10.1016/B978-0-12-820874-8.00001-4
Mohammed, M. A., Syeda, J. T. M., Wasan, K. M., & Wasan, E. K. (2017). An overview of chitosan nanoparticles and their application in non-parenteral drug delivery. Pharmaceutics, 9(4), 53. https://doi.org/10.3390/pharmaceutics9040053 DOI: https://doi.org/10.3390/pharmaceutics9040053
Grierosu, C., Calin, G., Damir, D., Marcu, C., Cernei, R., Zegan, G., Anistoroaei, D., Moscu, M., Carausu, E. M., Duceac, L. D., Dabija, M. G., Mitrea, G., Gutu, C., Bogdan Goroftei, E. R., & Eva, L. (2023). Development and Functionalization of a Novel Chitosan-Based Nanosystem for Enhanced Drug Delivery. Journal of Functional Biomaterials, 14(11), 538. https://doi.org/10.3390/jfb14110538 DOI: https://doi.org/10.3390/jfb14110538
Martins, A. F., Facchi, S. P., Follmann, H. D., et al. (2014). Antimicrobial activity of chitosan derivatives containing N quaternized moieties in its backbone: A review. International Journal of Molecular Sciences, 15(11), 20800–20832. https://doi.org/10.3390/ijms151120800 DOI: https://doi.org/10.3390/ijms151120800
Jiménez-Gómez, C. P., & Cecilia, J. A. (2020). Chitosan: A natural biopolymer with a wide and varied range of applications. Molecules, 25(17), 3981. https://doi.org/10.3390/molecules25173981 DOI: https://doi.org/10.3390/molecules25173981
Agnihotri, S. A., Mallikarjuna, N. N., & Aminabhavi, T. M. (2004). Recent advances in chitosan-based micro- and nanoparticles for drug delivery. Journal of Controlled Release, 100(1), 5–28. https://doi.org/10.1016/j.jconrel.2004.08.010 DOI: https://doi.org/10.1016/j.jconrel.2004.08.010
Illum, L. (1998). Chitosan and its use as a pharmaceutical excipient. Pharmaceutical Research, 15(9), 1326–1331. https://doi.org/10.1023/A:1011929016601 DOI: https://doi.org/10.1023/A:1011929016601
Elsabee, M. Z., & Abdou, E. S. (2013). Chitosan-based edible films and coatings: A review. Materials Science and Engineering C, 33(4), 1819–1841. https://doi.org/10.1016/j.msec.2013.01.010 DOI: https://doi.org/10.1016/j.msec.2013.01.010
Sogias, I. A., Williams, A. C., & Khutoryanskiy, V. V. (2008). Why is chitosan mucoadhesive? Biomacromolecules, 9(7), 1837–1842. https://doi.org/10.1021/bm800276d DOI: https://doi.org/10.1021/bm800276d
Bernkop-Schnürch, A., & Dünnhaupt, S. (2012). Chitosan-based drug delivery systems. European Journal of Pharmaceutics and Biopharmaceutics, 81(3), 463–469. https://doi.org/10.1016/j.ejpb.2012.04.007 DOI: https://doi.org/10.1016/j.ejpb.2012.04.007
Kean, T., & Thanou, M. (2010). Biodegradation, biodistribution, and toxicity of chitosan. Advanced Drug Delivery Reviews, 62(1), 3–11. https://doi.org/10.1016/j.addr.2009.09.004 DOI: https://doi.org/10.1016/j.addr.2009.09.004
Wunderlich, B. (2002). The measurement of the crystallinity of polymers by DSC. Polymer, 43, 3873–3878. https://doi.org/10.1016/S0032-3861(02)00235-5 DOI: https://doi.org/10.1016/S0032-3861(02)00235-5
Van Bavel, N., Issler, T., Pang, L., Anikovskiy, M., & Prenner, E. J. (2023). A Simple Method for Synthesis of Chitosan Nanoparticles with Ionic Gelation and Homogenization. Molecules, 28(11), 4328. https://doi.org/10.3390/molecules28114328 DOI: https://doi.org/10.3390/molecules28114328
Zaman, M., Butt, M. H., Siddique, W., Iqbal, M. O., Nisar, N., Mumtaz, A., Nazeer, H. Y., Alshammari, A., & Riaz, M. S. (2022). Fabrication of PEGylated Chitosan Nanoparticles Containing Tenofovir Alafenamide: Synthesis and Characterization. Molecules, 27(23), 8401. https://doi.org/10.3390/molecules27238401 DOI: https://doi.org/10.3390/molecules27238401
Jafernik, K., Kaczmarek, M., Buta, M., et al. (2023). Chitosan-based nanoparticles as effective drug delivery systems: A review. Molecules, 28(4), 1963. https://doi.org/10.3390/molecules28041963 DOI: https://doi.org/10.3390/molecules28041963
Borges, M. M. C., Teixeira, H. F., & Sousa, J. J. (2025). Controlled release of perillyl alcohol via pH-responsive polymeric carriers: Fitting to Korsmeyer–Peppas model. ACS Omega, 10, 1–11. https://doi.org/10.1021/acsomega.5c04817 DOI: https://doi.org/10.1021/acsomega.5c04817
El-Naggar, N. E. A., Shiha, A. M., Mahrous, H., et al. (2022). Green synthesis of chitosan nanoparticles, optimization, and characterization against biofilms. Scientific Reports, 12, 19869. https://doi.org/10.1038/s41598-022-24303-5 DOI: https://doi.org/10.1038/s41598-022-24303-5
Gutiérrez-Ruiz, S. C., Cortés, H., González-Torres, M., et al. (2024). Optimize the parameters for synthesis by ionic gelation, purification, and freeze-drying of chitosan-TPP nanoparticles for biomedical applications. Journal of Biological Engineering, 18, 12. https://doi.org/10.1186/s13036-024-00403-w DOI: https://doi.org/10.1186/s13036-024-00403-w
Yuan, Q., Shah, J., Hein, S., & Misra, R. D. K. (2010). Controlled and extended drug release behavior of chitosan-based nanoparticle carrier. Acta Biomaterialia, 6(3), 1140–1148. https://doi.org/10.1016/j.actbio.2009.08.027 DOI: https://doi.org/10.1016/j.actbio.2009.08.027
Conte, R., De Luca, I., Valentino, A., Cerruti, P., Pedram, P., Cabrera-Barjas, G., Moeini, A., & Calarco, A. (2023). Hyaluronic Acid Hydrogel Containing Resveratrol-Loaded Chitosan Nanoparticles as an Adjuvant in Atopic Dermatitis Treatment. Journal of Functional Biomaterials, 14(2), 82. https://doi.org/10.3390/jfb14020082 DOI: https://doi.org/10.3390/jfb14020082
Martín-Escaño, A., Barbuzano, C., Rodríguez-Díaz, J. M., & Pérez-Herrero, E. (2026). Statistical optimization of chitosan-based synthesis strategies to generate albumin nanoparticles. Drug Delivery and Translational Research. https://doi.org/10.1007/s13346-026-02046-4 DOI: https://doi.org/10.1007/s13346-026-02046-4
Filippov, S. K., Khusnutdinov, R., Murmiliuk, A., Inam, W., Zakharova, L. Ya., Zhang, H., & Khutoryanskiy, V. V. (2023). Dynamic light scattering and transmission electron microscopy in drug delivery: A roadmap for correct characterization of nanoparticles and interpretation of results. Materials Horizons, 10, 5354–5370. https://doi.org/10.1039/d3mh00717k DOI: https://doi.org/10.1039/D3MH00717K
Kumirska, J., Czerwicka, M., Kaczynski, Z., et al. (2010). Application of spectroscopic methods for structural analysis of chitin and chitosan. Marine Drugs, 8(5), 1567–1636. https://doi.org/10.3390/md8051567 DOI: https://doi.org/10.3390/md8051567
Gayathri, G., D’Souza, J. Q., & Sundaram, N. G. (2023). UV induced photocatalytic degradation of caffeine using TiO2–H-beta zeolite composite. Minerals, 13(4), 465. https://doi.org/10.3390/min13040465 DOI: https://doi.org/10.3390/min13040465
Bhawani, S. A., Fong, S. S., & Ibrahim, M. N. M. (2015). Spectrophotometric analysis of caffeine. International Journal of Analytical Chemistry, 2015, 170239. https://doi.org/10.1155/2015/170239 DOI: https://doi.org/10.1155/2015/170239
Herdiana, Y., Wathoni, N., Shamsuddin, S., & Muchtaridi, M. (2022). Drug release study of the chitosan-based nanoparticles. Heliyon, 8(1), e08674. https://doi.org/10.1016/j.heliyon.2021.e08674 DOI: https://doi.org/10.1016/j.heliyon.2021.e08674
Meynaud, S., Huet, G., Brulé, D., Gardrat, C., Poinssot, B., & Coma, V. (2023). Impact of UV irradiation on the chitosan bioactivity for biopesticide applications. Molecules, 28(13), 4954. https://doi.org/10.3390/molecules28134954 DOI: https://doi.org/10.3390/molecules28134954
Edwards, A. A., & Alexander, B. D. (2010). Organic applications of UV–visible absorption spectroscopy. In Encyclopedia of Spectroscopy and Spectrometry (2nd ed., pp. 2030–2039). https://doi.org/10.1016/B978-0-12-374413-5.00013-0 DOI: https://doi.org/10.1016/B978-0-12-374413-5.00013-0
Rajam, K., et al. (2013). Effect of caffeine–Zn2+ system in preventing corrosion of carbon steel in well water. Journal of Chemistry, 2013, 521951. https://doi.org/10.1155/2013/521951 DOI: https://doi.org/10.1155/2013/521951
Hamed, A., Ghareeb, D., Mohamed, T. M., Hamed, M., Nofal, M. S., & Gaber, M. (2023). Caffeine–folic acid-loaded chitosan nanoparticles combined with methotrexate as a novel HepG2 immunotherapy targeting adenosine A2A receptor downstream cascade. BMC Complementary Medicine and Therapies, 23, 384. https://doi.org/10.1186/s12906-023-04212-4 DOI: https://doi.org/10.1186/s12906-023-04212-4
Stefanowska, K., Woźniak, M., Majka, J., Sip, A., Mrówczyńska, L., Kozak, W., Dobrucka, R., & Ratajczak, I. (2023). Chitosan Films with Caffeine and Propolis as Promising and Ecofriendly Packaging Materials. Applied Sciences, 13(22), 12351. https://doi.org/10.3390/app132212351 DOI: https://doi.org/10.3390/app132212351
Woźniak, M., Gromadzka, K., & Kwaśniewska-Sip, P. (2022). Chitosan–caffeine formulation as an ecological preservative in wood protection. Wood Science and Technology, 56, 1851–1867. https://doi.org/10.1007/s00226-022-01426-6 DOI: https://doi.org/10.1007/s00226-022-01426-6
Kamburova, K., Boshkova, N., Radeva, T., Shipochka, M., & Boshkov, N. (2024). Chitosan–alginate nanocontainers with caffeine as green corrosion inhibitors for the protection of galvanized steel. Crystals, 14(7), 660. https://doi.org/10.3390/cryst14070660 DOI: https://doi.org/10.3390/cryst14070660
Lehmann, R., & Stowasser, K. (2007). The crystal structure of anhydrous beta-caffeine as determined from X-ray powder-diffraction data. Chemistry — A European Journal, 13(10), 2908–2911. https://doi.org/10.1002/chem.200600973 DOI: https://doi.org/10.1002/chem.200600973
Affes, S., Aranaz, I., Acosta Contreras, F. N., & Heras, Á. (2022). Physicochemical and biological properties of chitosan derivatives with varying molecular weight produced by chemical depolymerization. Biomass Conversion and Biorefinery, 14(3), 1–11. https://doi.org/10.1007/s13399-022-02662-3 DOI: https://doi.org/10.1007/s13399-022-02662-3
Song, W., Zhang, Q., Guan, Y., Li, W., Xie, S., Tong, J., Li, M., & Ren, L. (2022). Synthesis and characterization of porous chitosan/saccharomycetes adsorption microspheres. Polymers, 14(11), 2292. https://doi.org/10.3390/polym14112292 DOI: https://doi.org/10.3390/polym14112292
Khanmohammadi, M., Elmizadeh, H., & Ghasemi, K. (2015). Investigation of the size and morphology of chitosan nanoparticles used in a drug delivery system employing chemometric techniques. Iranian Journal of Pharmaceutical Research, 14(3), e125312. https://doi.org/10.22037/ijpr.2015.1761
Sarfraz, A., Simo, A., Fenger, R., Christen, W., Rademann, K., Panne, U., & Emmerling, F. (2012). Morphological diversity of caffeine on surfaces: Needles and hexagons. Crystal Growth & Design, 12, 583–588. https://doi.org/10.1021/cg101358q DOI: https://doi.org/10.1021/cg101358q
Milkova, V., & Goycoolea, F. M. (2020). Encapsulation of caffeine in polysaccharide oil-core nanocapsules. Colloid and Polymer Science, 298(8), 1035–1041. https://doi.org/10.1007/s00396-020-04653-0 DOI: https://doi.org/10.1007/s00396-020-04653-0
Dash, M., Chiellini, F., Ottenbrite, R. M., & Chiellini, E. (2011). Chitosan — A versatile semi-synthetic polymer in biomedical applications. Progress in Polymer Science, 36(8), 981–1014. https://doi.org/10.1016/j.progpolymsci.2011.02.001 DOI: https://doi.org/10.1016/j.progpolymsci.2011.02.001
Naskar, S., Kuotsu, K., & Sharma, S. (2018). Chitosan-based nanoparticles as drug delivery systems: a review on two decades of research. Journal of Drug Targeting, 27(4), 379–393. https://doi.org/10.1080/1061186x.2018.1512112 DOI: https://doi.org/10.1080/1061186X.2018.1512112
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Khusnibonu A. Ergasheva, Noira R. Vokhidova

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



