Rifampicin Loaded Chitosan-Based Nanoparticles: Optimization, Characterization, and Mucoadhesion
DOI:
https://doi.org/10.31489/2959-0663/1-26-4Keywords:
drug delivery, rifampicin, bioavailability, nanoparticles, chitosan, tuberculosis, anti-tuberculosis drugs, ionic gelation, mucoadhesion, mucinAbstract
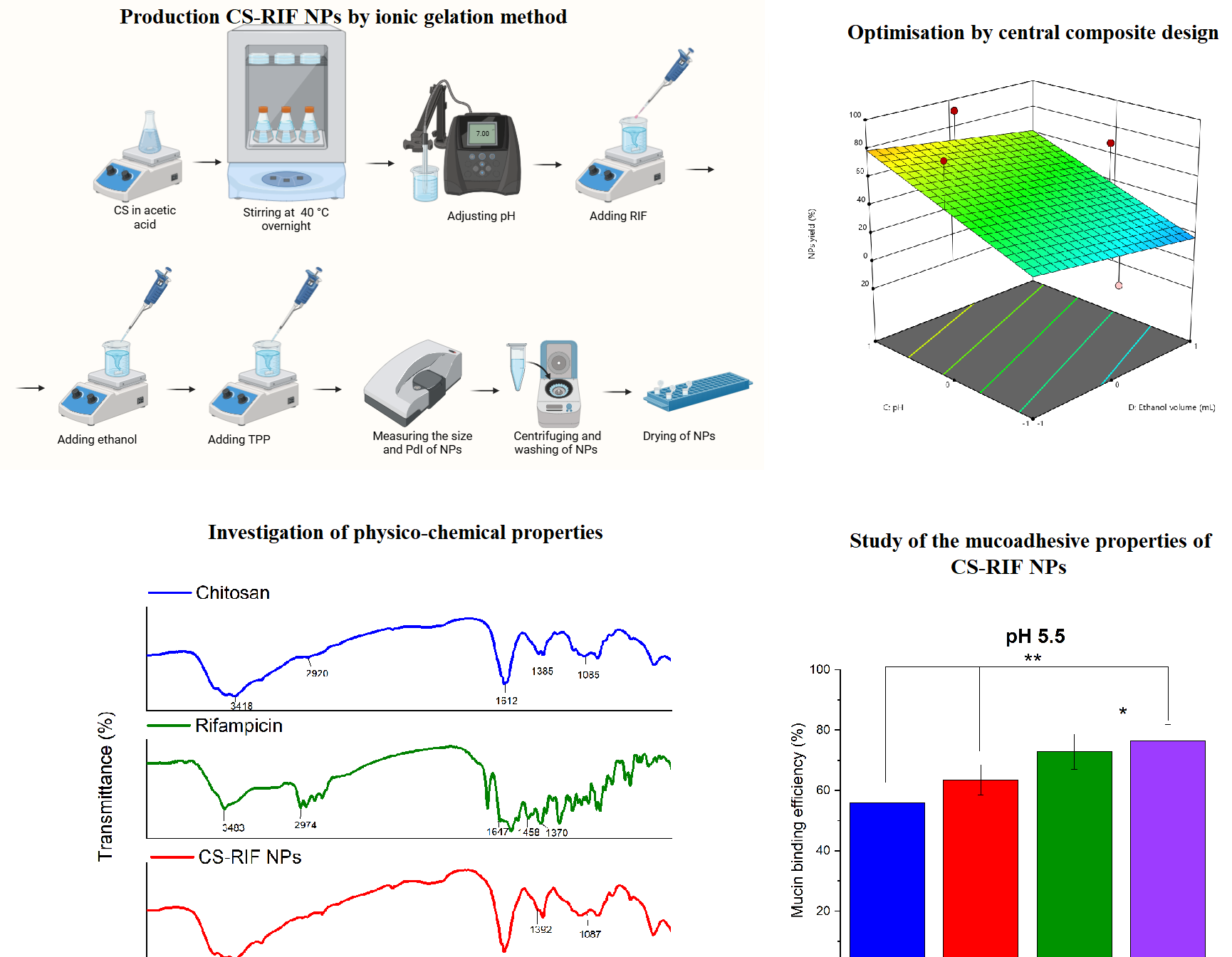
One of the important problems in modern pharmaceutical technology is the development of effective and safe drug delivery systems. In this regard, the development of nanostructures that deliver drugs in a targeted manner and increase their bioavailability is of particular importance. Biodegradable polymers form the basis of such systems. Among natural polysaccharides, chitosan deserves special attention. Colloidal particles made from chitosan, especially nanoparticle-based systems, increase the solubility of drugs and enable their effective delivery through the mucosal layer. This study aimed to prepare chitosan nanoparticles loaded with an anti-tuberculosis drug (rifampicin) using the ionotropic gelation method. A central composite design (CCD) was used to study the effects of chitosan concentration, rifampicin concentration, medium pH, and ethanol volume on particle size, polydispersity, and nanoparticle yield. The optimized nanoparticles were spherical in shape with an average particle size of 386±9 nm and a polydispersity index of 0.259±0.025. The rifampicin loading and nanoparticle yield of the optimized nanoparticles were 20 % and 71 %, respectively. The produced nanoparticles were analyzed using thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC), and the results showed no interaction between the drug and the polymer. Drug release from the polymer matrix was studied at different pH values stimulating the gastrointestinal tract. The mucoadhesive activity of rifampicin-loaded chitosan nanoparticles was investigated through the interaction with mucin in acetate buffer solution (pH 5.5) and phosphate buffer solution (pH 6.8). The results showed higher mucoadhesive activity in an acetate buffer solution.
References
World Health Organization. (2025). Global tuberculosis report 2025. Geneva: World Health Organization. Available at: https://www.who.int/publications/i/item/9789240116924
Carnero Canales, C. S., Marquez Cazorla, J. I., Marquez Cazorla, R. M., Roque-Borda, C. A., Polinário, G., Figueroa Banda, R. A., Sábio, R. M., Chorilli, M., Santos, H. A., & Pavan, F. R. (2024). Breaking barriers: The potential of nanosystems in antituberculosis therapy. Bioactive Materials, 39, 106–134. https://doi.org/10.1016/j.bioactmat.2024.05.013 DOI: https://doi.org/10.1016/j.bioactmat.2024.05.013
Yessentayeva, N. A., Galiyeva, A. R., Daribay, A. T., Sadyrbekov, D. T., Moustafine, R. I., & Tazhbayev, Y. M. (2024). Optimization of Polylactide-Co-Glycolide-Rifampicin Nanoparticle Synthesis, In Vitro Study of Mucoadhesion and Drug Release. Polymers, 16(17), 2466. https://doi.org/10.3390/polym16172466 DOI: https://doi.org/10.3390/polym16172466
Galiyeva, A.R., Tazhbayev, Ye.M., Zhumagaliyeva, T.S., & Daribay, A.Т. (2022). Encapsulation of Isoniazid in Polylactide-Co-Glycolide Nanoparticles by Nanoprecipitation. Bulletin of the University of Karaganda — Chemistry, 107(3), 208–217. https://doi.org/10.31489/2022Ch3/3-22-17 DOI: https://doi.org/10.31489/2022Ch3/3-22-17
Galiyeva, A. R., Tazhbayev, Ye. M., Zhumagaliyeva, T. S., Sadyrbekov, D. T., Kaikenov, D. A., Karimova, B. N., & Shokenova, S. S. (2022). Polylactide-co-glycolide nanoparticles immobilized with isoniazid: optimization using the experimental Taguchi method. Bulletin of the Karaganda University. “Chemistry” Series, 105(1), 69–77. https://doi.org/10.31489/2022ch1/69-77 DOI: https://doi.org/10.31489/2022Ch1/69-77
Galiyeva, A. R., Tazhbayev, Y. M., Yessentayeva, N. A., Daribay, A. T., Marsel, D. T., Sadyrbekov, D. T., Zhaparova, L. Zh., & Arystanova, Z. T. (2023). PEGylation of Albumin Nanoparticles Immobilized with the Anti-Tuberculosis Drug “Isoniazid.” Eurasian Journal of Chemistry, 28(2(110)). https://doi.org/10.31489/2959-0663/2-23-7 DOI: https://doi.org/10.31489/2959-0663/2-23-7
Sogias, I. A., Williams, A. C., & Khutoryanskiy, V. V. (2008). Why is Chitosan Mucoadhesive? Biomacromolecules, 9(7), 1837–1842. https://doi.org/10.1021/bm800276d DOI: https://doi.org/10.1021/bm800276d
Žigrayová, D., Mikušová, V., & Mikuš, P. (2024). Advances in Chitosan Derivatives: Preparation, Properties and Applications in Pharmacy and Medicine. Gels, 10(11), 701. https://doi.org/10.3390/gels10110701 DOI: https://doi.org/10.3390/gels10110701
Morin-Crini, N., Lichtfouse, E., Torri, G., & Crini, G. (2019). Fundamentals and Applications of Chitosan. Sustainable Agriculture Reviews 35, 49–123. https://doi.org/10.1007/978-3-030-16538-3_2 DOI: https://doi.org/10.1007/978-3-030-16538-3_2
Saputra, H. A., & Andreas. (2025). Chitosan and its biomedical applications: A review. Next Materials, 9, 101270. https://doi.org/10.1016/j.nxmate.2025.101270 DOI: https://doi.org/10.1016/j.nxmate.2025.101270
Herdiana, Y., Febrina, E., Nurhasanah, S., Gozali, D., Elamin, K. M., & Wathoni, N. (2024). Drug Loading in Chitosan-Based Nanoparticles. Pharmaceutics, 16(8), 1043. https://doi.org/10.3390/pharmaceutics16081043 DOI: https://doi.org/10.3390/pharmaceutics16081043
Jafernik, K., Ładniak, A., Blicharska, E., Czarnek, K., Ekiert, H., Wiącek, A. E., & Szopa, A. (2023). Chitosan-Based Nanoparticles as Effective Drug Delivery Systems—A review. Molecules, 28(4), 1963. https://doi.org/10.3390/molecules28041963 DOI: https://doi.org/10.3390/molecules28041963
Grewal, A. K., & Salar, R. K. (2024). Chitosan nanoparticle delivery systems: An effective approach to enhancing efficacy and safety of anticancer drugs. Nano TransMed, 3, 100040. https://doi.org/10.1016/j.ntm.2024.100040 DOI: https://doi.org/10.1016/j.ntm.2024.100040
Dago-Serry, Y., Maroulas, K. N., Tolkou, A. K., Kokkinos, N. C., & Kyzas, G. Z. (2024). How the chitosan structure can affect the adsorption of pharmaceuticals from wastewaters: An overview. Carbohydrate Polymer Technologies and Applications, 7, 100466. https://doi.org/10.1016/j.carpta.2024.100466 DOI: https://doi.org/10.1016/j.carpta.2024.100466
Gonciarz, W., Balcerczak, E., Brzeziński, M., Jeleń, A., Pietrzyk-Brzezińska, A. J., Narayanan, V. H. B., & Chmiela, M. (2025). Chitosan-based formulations for therapeutic applications. A recent overview. Journal of Biomedical Science, 32(1). https://doi.org/10.1186/s12929-025-01161-7 DOI: https://doi.org/10.1186/s12929-025-01161-7
Yang, C., Gao, S., Dagnæs-Hansen, F., Jakobsen, M., & Kjems, J. (2017). Impact of PEG Chain Length on the Physical Properties and Bioactivity of PEGylated Chitosan/siRNA Nanoparticles in Vitro and in Vivo. ACS Applied Materials & Interfaces, 9(14), 12203–12216. https://doi.org/10.1021/acsami.6b16556 DOI: https://doi.org/10.1021/acsami.6b16556
Picchi, V., Gobbi, S., Fattizzo, M., Zefelippo, M., & Faoro, F. (2021). Chitosan Nanoparticles Loaded with N-Acetyl Cysteine to Mitigate Ozone and Other Possible Oxidative Stresses in Durum Wheat. Plants, 10(4), 691. https://doi.org/10.3390/plants10040691 DOI: https://doi.org/10.3390/plants10040691
Rampino, A., Borgogna, M., Blasi, P., Bellich, B., & Cesàro, A. (2013). Chitosan nanoparticles: Preparation, size evolution and stability. International Journal of Pharmaceutics, 455(1–2), 219–228. https://doi.org/10.1016/j.ijpharm.2013.07.034 DOI: https://doi.org/10.1016/j.ijpharm.2013.07.034
Samprasit, W., Opanasopit, P., & Chamsai, B. (2021). Alpha‐mangostin and resveratrol, dual‐drugs‐loaded mucoadhesive thiolated chitosan‐based nanoparticles for synergistic activity against colon cancer cells. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 110(6), 1221–1233. Portico. https://doi.org/10.1002/jbm.b.34992 DOI: https://doi.org/10.1002/jbm.b.34992
Vieira, A. C. C., Chaves, L. L., Pinheiro, M., Lima, S. C., Neto, P. J. R., Ferreira, D., Sarmento, B., & Reis, S. (2021). Lipid nanoparticles coated with chitosan using a one-step association method to target rifampicin to alveolar macrophages. Carbohydrate Polymers, 252, 116978. https://doi.org/10.1016/j.carbpol.2020.116978 DOI: https://doi.org/10.1016/j.carbpol.2020.116978
Vieira, A. C. C., Chaves, L. L., Pinheiro, S., Pinto, S., Pinheiro, M., Lima, S. C., Ferreira, D., Sarmento, B., & Reis, S. (2018). Mucoadhesive chitosan-coated solid lipid nanoparticles for better management of tuberculosis. International Journal of Pharmaceutics, 536(1), 478–485. https://doi.org/10.1016/j.ijpharm.2017.11.071 DOI: https://doi.org/10.1016/j.ijpharm.2017.11.071
Yanat, M., & Schroën, K. (2021). Preparation methods and applications of chitosan nanoparticles; with an outlook toward reinforcement of biodegradable packaging. Reactive and Functional Polymers, 161, 104849. https://doi.org/10.1016/j.reactfunctpolym.2021.104849 DOI: https://doi.org/10.1016/j.reactfunctpolym.2021.104849
Jha, R., & Mayanovic, R. A. (2023). A Review of the Preparation, Characterization, and Applications of Chitosan Nanoparticles in Nanomedicine. Nanomaterials, 13(8), 1302. https://doi.org/10.3390/nano13081302 DOI: https://doi.org/10.3390/nano13081302
Divya, K., & Jisha, M. S. (2017). Chitosan nanoparticles preparation and applications. Environmental Chemistry Letters, 16(1), 101–112. https://doi.org/10.1007/s10311-017-0670-y DOI: https://doi.org/10.1007/s10311-017-0670-y
Rawal, T., Parmar, R., Tyagi, R. K., & Butani, S. (2017). Rifampicin loaded chitosan nanoparticle dry powder presents an improved therapeutic approach for alveolar tuberculosis. Colloids and Surfaces B: Biointerfaces, 154, 321–330. https://doi.org/10.1016/j.colsurfb.2017.03.044 DOI: https://doi.org/10.1016/j.colsurfb.2017.03.044
Patel, B. K., Parikh, R. H., & Aboti, P. S. (2013). Development of Oral Sustained Release Rifampicin Loaded Chitosan Nanoparticles by Design of Experiment. Journal of Drug Delivery, 2013, 1–10. https://doi.org/10.1155/2013/370938 DOI: https://doi.org/10.1155/2013/370938
Fan, W., Yan, W., Xu, Z., & Ni, H. (2012). Formation mechanism of monodisperse, low molecular weight chitosan nanoparticles by ionic gelation technique. Colloids and Surfaces B: Biointerfaces, 90, 21–27. https://doi.org/10.1016/j.colsurfb.2011.09.042 DOI: https://doi.org/10.1016/j.colsurfb.2011.09.042
Schnell, C. N., Galván, M. V., Solier, Y. N., Inalbon, M. C., Zanuttini, M. A., & Mocchiutti, P. (2021). High strength biobased films prepared from xylan/chitosan polyelectrolyte complexes in the presence of ethanol. Carbohydrate Polymers, 273, 118602. https://doi.org/10.1016/j.carbpol.2021.118602 DOI: https://doi.org/10.1016/j.carbpol.2021.118602
Shilova, S. V., Tret’yakova, A. Ya., & Barabanov, V. P. (2018). Association of Chitosan in Aqueous-Alcohol Solutions. Polymer Science, Series A, 60(2), 184–189. https://doi.org/10.1134/s0965545x1802013x DOI: https://doi.org/10.1134/S0965545X1802013X
Peyrovedin, H., Sajadian, S. A., Bahmanzade, S., Zomorodian, K., & Khorram, M. (2025). Studying the rifampin solubility in supercritical CO2 with/without co-solvent: Experimental data, modeling and machine learning approach. The Journal of Supercritical Fluids, 218, 106510. https://doi.org/10.1016/j.supflu.2024.106510 DOI: https://doi.org/10.1016/j.supflu.2024.106510
Galiyeva, A., Daribay, A., Zhumagaliyeva, T., Zhaparova, L., Sadyrbekov, D., & Tazhbayev, Y. (2023). Human Serum Albumin Nanoparticles: Synthesis, Optimization and Immobilization with Antituberculosis Drugs. Polymers, 15(13), 2774. https://doi.org/10.3390/polym15132774 DOI: https://doi.org/10.3390/polym15132774
Jogaiah, S., Mujtaba, A. G., Mujtaba, M., Archana, De Britto, S., Geetha, N., Belorkar, S. A., & Shetty, H. S. (2025). Chitosan-metal and metal oxide nanocomposites for active and intelligent food packaging; a comprehensive review of emerging trends and associated challenges. Carbohydrate Polymers, 357, 123459. https://doi.org/10.1016/j.carbpol.2025.123459 DOI: https://doi.org/10.1016/j.carbpol.2025.123459
Alehosseini, E., Shahiri Tabarestani, H., Kharazmi, M. S., & Jafari, S. M. (2022). Physicochemical, Thermal, and Morphological Properties of Chitosan Nanoparticles Produced by Ionic Gelation. Foods, 11(23), 3841. https://doi.org/10.3390/foods11233841 DOI: https://doi.org/10.3390/foods11233841
Onugwu, A. L., Attama, A. A., Nnamani, P. O., Onugwu, S. O., Onuigbo, E. B., & Khutoryanskiy, V. V. (2022). Development and optimization of solid lipid nanoparticles coated with chitosan and poly(2-ethyl-2-oxazoline) for ocular drug delivery of ciprofloxacin. Journal of Drug Delivery Science and Technology, 74, 103527. https://doi.org/10.1016/j.jddst.2022.103527 DOI: https://doi.org/10.1016/j.jddst.2022.103527
Alves, R., Reis, T. V. da S., Silva, L. C. C. da, Storpírtis, S., Mercuri, L. P., & Matos, J. do R. (2010). Thermal behavior and decomposition kinetics of rifampicin polymorphs under isothermal and non-isothermal conditions. Brazilian Journal of Pharmaceutical Sciences, 46(2), 343–351. https://doi.org/10.1590/s1984-82502010000200022 DOI: https://doi.org/10.1590/S1984-82502010000200022
Motiei, M., Pleno de Gouveia, L., Šopík, T., Vícha, R., Škoda, D., Císař, J., Khalili, R., Domincová Bergerová, E., Münster, L., Fei, H., Sedlařík, V., & Sáha, P. (2021). Nanoparticle-Based Rifampicin Delivery System Development. Molecules, 26(7), 2067. https://doi.org/10.3390/molecules26072067 DOI: https://doi.org/10.3390/molecules26072067
Kgoete, M. S., Mokgohloa, C. P., & Macevele, L. E. (2025). Synthesis of Chitosan Nanocomposite Materials Grafted with MWCNTs for the Removal of Tetracycline Pharmaceutical from Water Samples. Colloids and Interfaces, 9(5), 69. https://doi.org/10.3390/colloids9050069 DOI: https://doi.org/10.3390/colloids9050069
Herdiana, Y., Wathoni, N., Shamsuddin, S., & Muchtaridi, M. (2022). Drug release study of the chitosan-based nanoparticles. Heliyon, 8(1), e08674. https://doi.org/10.1016/j.heliyon.2021.e08674 DOI: https://doi.org/10.1016/j.heliyon.2021.e08674
Ghosh, R., Mondal, S., Mukherjee, D., Adhikari, A., Ahmed, S. A., Alsantali, R. I., Khder, A. S., Altass, H. M., Moussa, Z., Das, R., Bhattacharyya, M., & Pal, S. K. (2022). Oral drug delivery using a polymeric nanocarrier: chitosan nanoparticles in the delivery of rifampicin. Materials Advances, 3(11), 4622–4628. https://doi.org/10.1039/d2ma00295g DOI: https://doi.org/10.1039/D2MA00295G
England, C. G., Miller, M. C., Kuttan, A., Trent, J. O., & Frieboes, H. B. (2015). Release kinetics of paclitaxel and cisplatin from two and three layered gold nanoparticles. European Journal of Pharmaceutics and Biopharmaceutics, 92, 120–129. https://doi.org/10.1016/j.ejpb.2015.02.017 DOI: https://doi.org/10.1016/j.ejpb.2015.02.017
Pavaloiu, R.-D., Sha’at, F., Hlevca, C., Sha’at, M., Savoiu, G., & Osman, S. (2021). Evaluation of drug release kinetics from polymeric nanoparticles loaded with poorly water-soluble APIs. Ovidius University Annals of Chemistry, 32(2), 132–136. https://doi.org/10.2478/auoc-2021-0020 DOI: https://doi.org/10.2478/auoc-2021-0020
Jahangiri, S., Amirkhani, L., Akbarzadeh, A., & Hajimohammadi, R. (2023). Study of the Release Kinetic and the Diffusion Coefficient of Doxorubicin-Chrysin Coated with Fe3O4 and Polycaprolactone-Polyethylene glycol Copolymers. Iranian Journal of Chemistry and Chemical Engineering, 42(5), 1436–1446.
Khutoryanskiy, V. V. (2018). Beyond PEGylation: Alternative surface-modification of nanoparticles with mucus-inert biomaterials. Advanced Drug Delivery Reviews, 124, 140–149. https://doi.org/10.1016/j.addr.2017.07.015 DOI: https://doi.org/10.1016/j.addr.2017.07.015
Kaldybekov, D. B., Shatabayeva, E. O., Polatkhan, A. A., Tuleyeva, R. N., Irmukhametova, G. S., & Khutoryanskiy, V. V. (2024). Development and Investigation of Mucoadhesive Polymers Based on Chitosan for Intravesical Therapy. Eurasian Journal of Chemistry, 29(4(116)), 13–21. https://doi.org/10.31489/2959-0663/4-24-2 DOI: https://doi.org/10.31489/2959-0663/4-24-2
Phuong Ta, L., Bujna, E., Kun, S., Charalampopoulos, D., & Khutoryanskiy, V. V. (2021). Electrosprayed mucoadhesive alginate-chitosan microcapsules for gastrointestinal delivery of probiotics. International Journal of Pharmaceutics, 597, 120342. https://doi.org/10.1016/j.ijpharm.2021.120342 DOI: https://doi.org/10.1016/j.ijpharm.2021.120342
Tousian, B., & Khosravi, A. R. (2023). Chitosan-based pulmonary particulate systems for anticancer and antiviral drug carriers: A promising delivery for COVID-19 vaccines. Results in Chemistry, 6, 101146. https://doi.org/10.1016/j.rechem.2023.101146 DOI: https://doi.org/10.1016/j.rechem.2023.101146
Zacaron, T. M., Silva, M. L. S. e, Costa, M. P., Silva, D. M. e, Silva, A. C., Apolônio, A. C. M., Fabri, R. L., Pittella, F., Rocha, H. V. A., & Tavares, G. D. (2023). Advancements in Chitosan-Based Nanoparticles for Pulmonary Drug Delivery. Polymers, 15(18), 3849. https://doi.org/10.3390/polym15183849 DOI: https://doi.org/10.3390/polym15183849
Rasul, R. M., Tamilarasi Muniandy, M., Zakaria, Z., Shah, K., Chee, C. F., Dabbagh, A., Rahman, N. A., & Wong, T. W. (2020). A review on chitosan and its development as pulmonary particulate anti-infective and anti-cancer drug carriers. Carbohydrate Polymers, 250, 116800. https://doi.org/10.1016/j.carbpol.2020.116800 DOI: https://doi.org/10.1016/j.carbpol.2020.116800
Vandal, O. H., Nathan, C. F., & Ehrt, S. (2009). Acid Resistance in Mycobacterium tuberculosis. Journal of Bacteriology, 191(15), 4714–4721. https://doi.org/10.1128/jb.00305-09 DOI: https://doi.org/10.1128/JB.00305-09
Nyberg, K., Johansson, U., Rundquist, I., & Camner, P. (1989). Estimation of pH in Individual Alveolar Macrophage Phagolysosomes. Experimental Lung Research, 15(4), 499–510. https://doi.org/10.3109/01902148909069614 DOI: https://doi.org/10.3109/01902148909069614
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Yerkeblan M. Tazhbayev, Aldana R. Galiyeva, Ulpan Y. Syrymova, Lyazzat Zh. Zhaparova, Tolkyn S. Zhumagaliyeva

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



