Structural Characteristics, Stability, and Anticoagulant Activity of Bombyx mori Chitosan Sulfate
DOI:
https://doi.org/10.31489/2959-0663/1-26-5Keywords:
chitosan sulfate, Bombyx mori, degree of substitution, IR spectroscopy, X-ray structural analysis, water sorption, rheology, anticoagulant activityAbstract
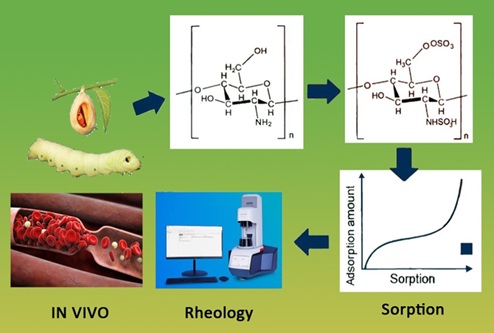
Chitosan sulfate (CS) is a promising polyelectrolyte for biomedical applications due to its anticoagulant properties. However, the relationship between the synthesis conditions of CS derived from Bombyx mori silkworm chitosan, its structural characteristics, and its biological activity remains insufficiently studied. CS samples with varying degrees of substitution (DS) were synthesized via sulfation with chlorosulfonic acid at different temperatures (50, 60, and 80 °C). The structure was characterized using FTIR spectroscopy and X-ray diffraction analysis. The physicochemical properties were evaluated through water vapor sorption, rheology, and solution stability tests conducted over 30 days. The anticoagulant activity of CS was assessed in vivo using a rabbit model of hypercholesterolemia. Successful sulfation resulted in DS values ranging from 0.96 to 1.21, leading to significant amorphization of the polymer structure. All CS samples exhibited high hydrophilicity with sigmoid-shaped sorption isotherms and demonstrated typical polyelectrolyte rheological behavior. Aqueous solutions of CS remained stable throughout the observation period. Importantly, the CS sample with the highest DS (1.21) showed the most pronounced anticoagulant effect, reducing platelet aggregation by 20 % compared with the heparin control group. The sulfation temperature is a key parameter determining the DS and, consequently, the properties of Bombyx mori chitosan sulfate. The derivative with DS = 1.21 demonstrates anticoagulant activity comparable to heparin, highlighting its potential as a bioactive material. Further research should focus on elucidating the precise molecular mechanisms of its anticoagulant action and evaluating its long-term biocompatibility and efficacy in vivo.
References
Wang, W., Meng, Q., Li, Q., Liu, J., Zhou, M., Jin, Z., & Zhao, K. (2020). Chitosan Derivatives and Their Application in Biomedicine. International Journal of Molecular Sciences, 21(2), 487. https://doi.org/10.3390/ijms21020487 DOI: https://doi.org/10.3390/ijms21020487
Žigrayová, D., Mikušová, V., & Mikuš, P. (2024). Advances in chitosan derivatives: Preparation, properties, and applications in pharmacy and medicine. Gels, 10(11), 701. https://doi.org/10.3390/gels10110701 DOI: https://doi.org/10.3390/gels10110701
Hemmami, H., Ben Amor, A., Zeghoud, S., Ahmed, S., & Alnazza Alhamad, A. (2023). Chitosan, its derivatives, sources, preparation methods, and applications: A review. Journal of the Turkish Chemical Society Section A: Chemistry, 11(1), 341–354. https://doi.org/10.18596/jotcsa.1336313 DOI: https://doi.org/10.18596/jotcsa.1336313
Sashiwa, H., Kawasaki, N., Nakayama, A., et al. (2003). Chemical modification of chitosan. Part 15: Synthesis of novel chitosan derivatives by substitution of hydrophilic amine using N-carboxyethylchitosan ethyl ester as an intermediate. Carbohydrate Research, 338(6), 557–561. https://doi.org/10.1016/S0008-6215(02)00492-5 DOI: https://doi.org/10.1016/S0008-6215(02)00492-5
Chen, Q., Qi, Y., Jiang, Y., Quan, W., Luo, H., Wu, K., Li, S., & Ouyang, Q. (2022). Progress in Research of Chitosan Chemical Modification Technologies and Their Applications. Marine Drugs, 20(8), 536. https://doi.org/10.3390/md20080536 DOI: https://doi.org/10.3390/md20080536
Wang, Z., Zheng, L., Li, C., Wu, S., & Xiao, Y. (2017). Preparation and antimicrobial activity of sulfopropyl chitosan in an ionic liquid aqueous solution. Journal of Applied Polymer Science, 134(15), 44989. https://doi.org/10.1002/app.44989 DOI: https://doi.org/10.1002/app.44989
Petrova, Y. S., Neudachina, L. K., Mekhaev, A. V., & Pestov, A. V. (2014). Simple synthesis and chelation capacity of N-(2-sulfoethyl)chitosan, a taurine derivative. Carbohydrate Polymers, 112, 462–468. https://doi.org/10.1016/j.carbpol.2014.06.028 DOI: https://doi.org/10.1016/j.carbpol.2014.06.028
Pestov, A. V., Petrova, Y. S., Bukharova, A. V., Neudachina, L. K., et al. (2013). Synthesis in a gel and sorption properties of N-2-sulfoethyl chitosan. Russian Journal of Applied Chemistry, 86(2), 269–272. https://doi.org/10.1134/S1070427213020225 DOI: https://doi.org/10.1134/S1070427213020225
Petrova, V. А., Chernyakov, D. D., Moskalenko, Y. E., Gasilova, E. R., Strelina, I. А., et al. (2017). O,N-(2-sulfoethyl)chitosan: Synthesis and properties of solutions and films. Carbohydrate Polymers, 157, 866–874. https://doi.org/10.1016/j.carbpol.2016.10.058 DOI: https://doi.org/10.1016/j.carbpol.2016.10.058
Petrova, Y. S., Pestov, A. V., Usoltseva, M. K., & Neudachina, L. K. (2015). Selective adsorption of silver(I) ions over copper(II) ions on a sulfoethyl derivative of chitosan. Journal of Hazardous Materials, 299, 696–701. https://doi.org/10.1016/j.jhazmat.2015.08.001 DOI: https://doi.org/10.1016/j.jhazmat.2015.08.001
Pestov, A. V., Koryakova, O. V., Leonidov, I. I., & Yatluk, Y. G. (2010). Gel-synthesis, structure, and properties of sulfur-containing chitosan derivatives. Russian Journal of Applied Chemistry, 83(5), 787–794. https://doi.org/10.1134/S1070427210050058 DOI: https://doi.org/10.1134/S1070427210050058
Petrova, Y. S., Pestov, A. V., & Neudachina, L. K. (2016). Removal of metal ions in fixed bed from multicomponent solutions using N-(2-sulfoethyl)chitosan-based sorbents. Separation Science and Technology, 51(9), 1437–1445. https://doi.org/10.1080/01496395.2016.1157085 DOI: https://doi.org/10.1080/01496395.2016.1157085
Ginting, N. H. (2004). Synthesis of chitosan sulfate through sulfonation reaction of chitosan with chlorosulfonic acid in solvent N,N-dimethylformamide. Journal of Chemical Natural Resources, 4(1), 1–8. https://doi.org/10.32734/jcnar.v4i1.9353 DOI: https://doi.org/10.32734/jcnar.v4i1.9353
Sun, Z., Shi, C., Wang, X., Fang, Q., & Huang, J. (2017). Synthesis, characterization, and antimicrobial activities of sulfonated chitosan. Carbohydrate Polymers, 155, 321–328. https://doi.org/10.1016/j.carbpol.2016.08.069 DOI: https://doi.org/10.1016/j.carbpol.2016.08.069
Xie, W., Xu, P., & Liu, Q. (2001). Antioxidant activity of water-soluble chitosan derivatives. Bioorganic & Medicinal Chemistry Letters, 11(13), 1699–1701. https://doi.org/10.1016/S0960-894X(01)00285-2 DOI: https://doi.org/10.1016/S0960-894X(01)00285-2
Holme, K. R., & Perlin, A. S. (1997). Chitosan N-sulfate: A water-soluble polyelectrolyte. Carbohydrate Research, 302(1–2), 7–12. https://www.google.com/search? q=https://doi.org/10.1016/S0008-6215(97)00117-1 DOI: https://doi.org/10.1016/S0008-6215(97)00117-1
Qin, C., Li, H., Xiao, Q., Liu, Y., & Zhu, J. (2006). Water-solubility of chitosan and its antimicrobial activity. Carbohydrate Polymers, 63(3), 367–374. https://doi.org/10.1016/j.carbpol.2005.09.023 DOI: https://doi.org/10.1016/j.carbpol.2005.09.023
Seidi, F., Heshmati, P., Galehdari, S., Tajehmiri, A., & Salimi, H. (2016). Synthesis of water-soluble quaternary chitosan derivative via protection-deprotection strategy and investigation of its antibacterial effect. Polymer Science, Series B, 58(3), 341–346. https://doi.org/10.1134/S1560090416030143 DOI: https://doi.org/10.1134/S1560090416030143
Chouljenko, A., Chotiko, A., Reyes, V., Alfaro, L., Liu, C., et al. (2016). Application of water-soluble chitosan to shrimp for quality retention. LWT — Food Science and Technology, 74, 571–579. https://doi.org/10.1016/j.lwt.2016.08.024 DOI: https://doi.org/10.1016/j.lwt.2016.08.024
Wu, S. (2016). Preparation of water-soluble chitosan and its suppressive effect on the denaturation of scallop adductor muscle myofibrillar protein during frozen storage. Journal of Aquatic Food Product Technology, 25(8), 1368–1374. https://doi.org/10.1080/10498850.2015.1077299 DOI: https://doi.org/10.1080/10498850.2015.1077299
Hou, J. W., Qian, L., Kou, J. M., Zhang, C. W., Jia, X. J., et al. (2015). Effect of water-soluble chitosan on the osteoblast function in MC3T3-E1 cells. International Journal of Biological Macromolecules, 72, 1041–1043. https://doi.org/10.1016/j.ijbiomac.2014.10.012 DOI: https://doi.org/10.1016/j.ijbiomac.2014.10.012
Sahariah, P., & Masson, M. (2017). Antimicrobial chitosan and chitosan derivatives: A review of the structure–activity relationship. Biomacromolecules, 18(11), 3846–3868. https://doi.org/10.1021/acs.biomac.7b01058 DOI: https://doi.org/10.1021/acs.biomac.7b01058
Martins, A. F., Facchi, S. P., Follmann, H. D., Pereira, A. G., Rubira, A. F., et al. (2014). Antimicrobial activity of chitosan derivatives containing N-quaternized moieties in its backbone: A review. International Journal of Molecular Sciences, 15(11), 20800–20832. https://doi.org/10.3390/ijms151120800 DOI: https://doi.org/10.3390/ijms151120800
Seedevi, P., Moovendhan, M., Vairamani, S., & Shanmugam, A. (2017). Evaluation of antioxidant activities and chemical analysis of sulfated chitosan from Sepia prashadi. International Journal of Biological Macromolecules, 103, 519–529. https://doi.org/10.1016/j.ijbiomac.2017.03.012 DOI: https://doi.org/10.1016/j.ijbiomac.2017.03.012
Vino, A. B., Ramasamy, P., Shanmugam, V., & Shanmugam, A. (2012). Extraction, characterization, and in vitro antioxidative potential of chitosan and sulfated chitosan from cuttlebone of Sepia aculeata Orbigny, 1848. Asian Pacific Journal of Tropical Biomedicine, 2(1), S334–S341. https://doi.org/10.1016/S2221-1691(12)60184-1 DOI: https://doi.org/10.1016/S2221-1691(12)60184-1
Vongchan, P., Sajomsang, W., Kasinrerk, W., Subyen, D., & Kongtaweleri, P. (2002). Anticoagulant activities of the chitosan polysulfate synthesized from marine crab shell by semi-heterogeneous conditions. Carbohydrate Research, 337(13), 1239–1242. https://doi.org/10.1016/S0008-6215(02)00098-8 DOI: https://doi.org/10.1016/S0008-6215(02)00098-8
Dinoro, J., Maher, M., Talebian, S., Jafarkhani, M., Mehrali, M., Orive, G., Foroughi, J., et al. (2019). Sulfated polysaccharide-based scaffolds for orthopaedic tissue engineering. Biomaterials, 214, 119214. https://doi.org/10.1016/j.biomaterials.2019.05.025 DOI: https://doi.org/10.1016/j.biomaterials.2019.05.025
Zhang, M., Haga, A., Sekiguchi, H., & Hirano, S. (2000). Structure of insect chitin isolated from beetle larva cuticle and silkworm (Bombyx mori) pupa exuvia. International Journal of Biological Macromolecules, 27(1), 99–105. https://doi.org/10.1016/S0141-8130(99)00123-3 DOI: https://doi.org/10.1016/S0141-8130(99)00123-3
Milusheva, R., & Rashidova, S. Sh. (2019). Bombyx mori chitosan nanoparticles: Synthesis and properties. Open Journal of Organic Polymer Materials, 9(4), 63–73. https://doi.org/10.4236/ojopm.2019.94004 DOI: https://doi.org/10.4236/ojopm.2019.94004
Vokhidova, N. R., Mamasoliyev, U. M., Yugay, S. M., et al. (2023). Synthesis and study of the structure of N-succinyl chitosan Bombyx mori and their biological applications. Polymer Bulletin, 80, 12907–12921. https://doi.org/10.1007/s00289-023-04680-1 DOI: https://doi.org/10.1007/s00289-023-04680-1
Milusheva, R. Y., & Rashidova, S. Sh. (2017). Bioactive properties of nanochitosan Bombyx mori. Polymer Science, Series C, 59(1), 29–34. https://doi.org/10.1134/S1811238217010088 DOI: https://doi.org/10.1134/S1811238217010088
Milusheva, R. Y., & Rashidova, S. Sh. (2022). Obtaining chitosan nanoparticles from Bombyx mori. Russian Chemical Bulletin, 71(2), 232–239. https://doi.org/10.1007/s11172-022-3402-9 DOI: https://doi.org/10.1007/s11172-022-3402-9
Jayakumar, R., Nwe, N., Tokura, S., & Tamura, H. (2007). Sulfated chitin and chitosan as novel biomaterials. International Journal of Biological Macromolecules, 40(3), 175–181. https://doi.org/10.1016/j.ijbiomac.2006.06.021 DOI: https://doi.org/10.1016/j.ijbiomac.2006.06.021
Dimassi, S., Tabary, N., Chai, F., Blanchemain, N., & Martel, B. (2018). Sulfonated and sulfated chitosan derivatives for biomedical applications: A review. Carbohydrate Polymers, 202, 382–396. https://doi.org/10.1016/j.carbpol.2018.09.011 DOI: https://doi.org/10.1016/j.carbpol.2018.09.011
Rakhmanova, V. N., Nud’ga, L. A., Milusheva, R. Y., et al. (2009). Determination of the degree of sulfation of Bombyx mori chitosan by conductometric titration. Russian Journal of Applied Chemistry, 82(12), 2192–2196. https://doi.org/10.1134/S1070427209120192 DOI: https://doi.org/10.1134/S1070427209120192
Pokharkar, V., Dhar, S., Bhumkar, D., et al. (2009). Acute and subacute toxicity studies of chitosan-reduced gold nanoparticles: A novel carrier for therapeutic agents. Journal of Biomedical Nanotechnology, 5(2), 233–239. https://doi.org/10.1166/jbn.2009.1027 DOI: https://doi.org/10.1166/jbn.2009.1027
Hirano, S., Kinugawa, J., & Nishioka, A. (1986). Sulfated derivatives of chitosan and their characterization with respect to biological activity. In R. Muzzarelli, C. Jeuniaux, & G.W. Gooday (Eds.), Chitin in Nature and Technology (pp. 461–468). Plenum Press. https://doi.org/10.1007/978-1-4613-2167-5_54 DOI: https://doi.org/10.1007/978-1-4613-2167-5_54
Furman, R. H., & Robinson, C.W. Jr. (1961). Hypocholesterolemic agents. Medical Clinics of North America, 45(4), 935–959. https://doi.org/10.1016/S0025-7125(16)33854-8 DOI: https://doi.org/10.1016/S0025-7125(16)33854-8
Figueroa, F. A., Abdala-Díaz, R. T., Pérez, C., Casas-Arrojo, V., Nesic, A., Tapia, C., Durán, C., Valdes, O., Parra, C., Bravo-Arrepol, G., Soto, L., Becerra, J., & Cabrera-Barjas, G. (2022). Sulfated Polysaccharide Extracted from the Green Algae Codium bernabei: Physicochemical Characterization and Antioxidant, Anticoagulant and Antitumor Activity. Marine Drugs, 20(7), 458. https://doi.org/10.3390/md20070458 DOI: https://doi.org/10.3390/md20070458
Ergashev, K.H., Vokhidova, N.R., Rashidova S.Sh. (2022) Synthesis and application of Chitosan hydroxyapatite: Review. Progress on Chemistry and Application of Chitin and its Derivatives, 27, 5–34. https://doi.org/10.15259/PCACD.27.001. DOI: https://doi.org/10.15259/PCACD.27.001
Garcia, J., & Schultz, L. D. (2016). Determination of sulfate by conductometric titration: An undergraduate laboratory experiment. Journal of Chemical Education, 93(5), 910–914. https://doi.org/10.1021/acs.jchemed.5b00941 DOI: https://doi.org/10.1021/acs.jchemed.5b00941
Smith, B. (2018). Infrared Spectral Interpretation. CRC Press. https://doi.org/10.1201/9780203750841 DOI: https://doi.org/10.1201/9780203750841
Yugay, S. M., Shakhabutdinov, S. S., Atakhanov, A. A., & Rashidova, S. Sh. (2019). IR-Fourier spectroscopic research of Bombyx mori chitosan and its derivatives. Chemical Journal of Kazakhstan, 254–265. https://chemjournal.kz/index.php/journal/article/view/224
Duarte, M. L., Ferreira, M. C., Marvão, M. R., & Rocha, J. (2002). An optimised method to determine the degree of acetylation of chitin and chitosan by FTIR spectroscopy. International Journal of Biological Macromolecules, 31(1–3), 1–8. https://doi.org/10.1016/s0141-8130(02)00039-9 DOI: https://doi.org/10.1016/S0141-8130(02)00039-9
Clark, G. L., & Smith, A. F. (1936). X-ray diffraction studies of chitin, chitosan and derivatives. Journal of Physical Chemistry, 40(7), 863–879. https://doi.org/10.1021/j150376a001 DOI: https://doi.org/10.1021/j150376a001
Tager, A. A. (1972). Physical chemistry of polymers. Mir Publishers. Moscow.
Gregg S.J. Sing K.S.W. (1982). Adsorption, Surface Area, and Porosity, 2nd ed., Academic Press, New York.
Poyet, S., Trentin, K., & Amblard, E. (2016). The use of sorption balance for the characterization of the water retention curve of cement-based materials. Journal of Advanced Concrete Technology, 14(7), 354–367. https://doi.org/10.3151/jact.14.354 DOI: https://doi.org/10.3151/jact.14.354
Mali, S., Sakanaka, L. S., Yamashita, F., & Grossmann, M. V. E. (2005). Water sorption and mechanical properties of cassava starch films and their relation to plasticizing effect. Carbohydrate Polymers, 60(3), 283–289. https://doi.org/10.1016/j.carbpol.2005.01.003 DOI: https://doi.org/10.1016/j.carbpol.2005.01.003
Mezger, T. (2020). The rheology handbook: For users of rotational and oscillatory rheometers. European Coatings. https://doi.org/10.1515/arh-2002-0029 DOI: https://doi.org/10.1515/9783748603702
Hanna Instruments. (n.d.). User manual for EC 215 conductivity meter. https://www.hannainst.com
Korshak, V. V., & Vinogradova, S. V. (1968). Dependence of thermal stability of polymers on their chemical structure. Russian Chemical Reviews, 37(11), 885. https://doi.org/10.1070/RC1968v037n11ABEH001712 DOI: https://doi.org/10.1070/RC1968v037n11ABEH001712
Masuelli, M. A. (2014). Mark-Houwink parameters for aqueous-soluble polymers and biopolymers at various temperatures. Journal of Polymer and Biopolymer Physics Chemistry, 2(2), 37–43. https://pubs.sciepub.com/jpbpc/2/2/2/
American Polymer Standards Corporation. (2022). Mark-Houwink parameters for polymer.
Brandrup, J., Immergut, E. H. Grulke, E. A. (2003). Polymer Handbook (4th ed.). New York: Wiley.
Vogel, H. G. (Ed.). (2008). Drug Discovery and Evaluation. https://doi.org/10.1007/978-3-540-70995-4 DOI: https://doi.org/10.1007/978-3-540-70995-4
Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. (2010). Official Journal of the European Union, L 276/33. https://eur-lex.europa.eu/eli/dir/2010/63/oj/eng
Znamenskaya, Y., Sotres, J., Engblom, J., Arnebrant, T., & Kocherbitov, V. (2012). Effect of hydration on structural and thermodynamic properties of pig gastric and bovine submaxillary gland mucins. The Journal of Physical Chemistry B, 116(16), 5047–5055. https://doi.org/10.1021/jp212495t DOI: https://doi.org/10.1021/jp212495t
Digaitis, R., Falkman, P., Oltner, V., Briggner, L. E., & Kocherbitov, V. (2022). Hydration and dehydration induced changes in porosity of starch microspheres. Carbohydrate Polymers, 291, 119542. https://doi.org/10.1016/j.carbpol.2022.119542 DOI: https://doi.org/10.1016/j.carbpol.2022.119542
Argatov, I., & Kocherbitov, V. (2021). An empirical model for sorption by glassy polymers: An assessment of thermodynamic parameters. Polymer Testing, 99, 107220. https://doi.org/10.1016/j.polymertesting.2021.107220 DOI: https://doi.org/10.1016/j.polymertesting.2021.107220
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Vazira N. Rakhmanova, Svetlana M. Yugay , Rakiya Yu. Milusheva, Sirojiddin Sh. Shakhabutdinov, Nurbek Sh. Ashurov, Khumoyunmirzo A. Gulomjonov, Abdumutolib A. Atakhanov, Sayyora Sh. Rashidova

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



