Engineering Biocompatible Goethite Nanoparticles: Microstructural Tuning through Controlled Ferrihydrite Conversion Routes
DOI:
https://doi.org/10.31489/2959-0663/1-26-9Keywords:
goethite nanoparticles, ferrihydrite precursor, aging, ultrasonic-assisted conversion, hydrothermal crystallization, post-synthesis transformation, varying the synthesis conditions, crystal structureAbstract
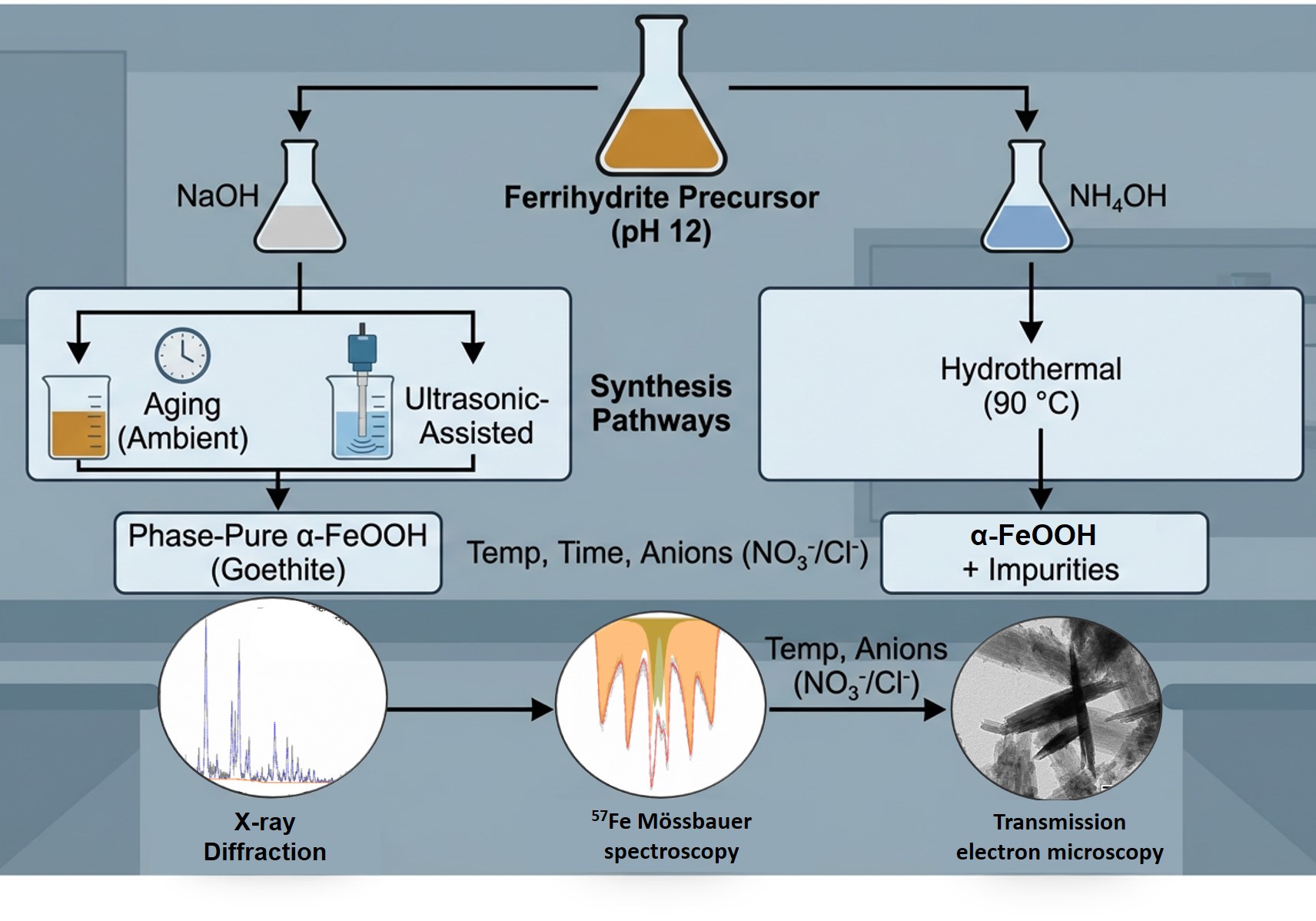
The goethite (α-FeOOH) nanoparticles (NPs) are highly attractive material with a broad spectrum of applications, including biomedicine thanks to high thermodynamical stability. The formation and strict control of the phase-pure goethite remains a challenging task due to the high sensitivity of the particles to the synthesis conditions. This study presents a comparative analysis of three distinct post-synthesis transformation pathways for preparing goethite NPs from ferrihydrite suspensions: aging, ultrasonic-assisted conversion, and hydrothermal crystallization. We observed that ferrihydrite NPs precursor generated with NaOH facilitated the formation of phase-pure goethite through both aging and ultrasonic transformation under ambient conditions. In contrast, at 90 °C promoted the formation of larger crystals, often accompanied by minor phase impurities. While the Cl- influenced structural evolution, our study underscores the dominant role of synthesis parameters, particularly thermal and temporal control, in dictating the phase selectivity and morphological uniformity of goethite. Notably, aging-based syntheses, without thermal or ultrasonic enhancement, consistently yielded homogeneous, monophasic crystals exhibiting complete sorption (with partition coefficients approaching). Comprehensive characterization of the synthesized materials was performed using X-ray diffraction, 57Fe Mössbauer spectroscopy, and transmission electronic microscopy, enabling a detailed assessment of crystallinity, phase purity, and morphological features.
References
Martina, M. R., Zoli, L., Sani, E. (2022). Synthesis and characterization of goethite (α-FeOOH) magnetic nanofluids. International Journal of Thermofluids, 15, 100169. https://doi.org/10.1016/j.ijft.2022.100169 DOI: https://doi.org/10.1016/j.ijft.2022.100169
Kubiak, A., Voronkina, A., & Jesionowski, T. (2023). Creation of a 3D Goethite–Spongin Composite Using an Extreme Biomimetics Approach. Biomimetics, 8, 533. https://doi.org/10.3390/biomimetics8070533 DOI: https://doi.org/10.3390/biomimetics8070533
Nowacki, K., Kubiak, A., & Jesionowski, T. (2024). 3D Spongin Scaffolds as Templates for Electro-Assisted Deposition of Selected Iron Oxides. Biomimetics, 9, 387. https://doi.org/10.3390/biomimetics9070387 DOI: https://doi.org/10.3390/biomimetics9070387
Shi, Z., Zachara, J.M., & Fredrickson, J.K. (2012). Redox Reactions of Reduced Flavin Mononucleotide (FMN), Riboflavin (RBF), and Anthraquinone-2,6-disulfonate (AQDS) with Ferrihydrite and Lepidocrocite. Environmental Science & Technology, 46 (21), 11644–11652. https://doi.org/10.1021/es301544b DOI: https://doi.org/10.1021/es301544b
Zhourui, L., Manisha, M., Yichao, W., Qiaoyun, H., Peng C. (2021). Increased particle size of goethite enhances the antibacterial effect on human pathogen Escherichia coli O157:H7: A Raman spectroscopic study. Journal of Hazardous Materials, 405, 124174. https://doi.org/10.1016/j.jhazmat.2020.124174 DOI: https://doi.org/10.1016/j.jhazmat.2020.124174
Kosei, I., Eiji, A., Oluyomi, S. A. & (2024). Goethite and Hematite Nanoparticles Show Promising Anti-Toxoplasma Properties. Pharmaceutics, 16 (3), 413. https://doi.org/10.3390/pharmaceutics16030413 DOI: https://doi.org/10.3390/pharmaceutics16030413
Dulnee, S., Scheinost, A.C. (2014). Surface Reaction of SnII on Goethite (α-FeOOH): Surface Complexation, Redox Reaction, Reductive Dissolution, and Phase Transformation. Environmental Science & Technology, 48 (16), 9341–9348. https://doi.org/10.1021/es501923z DOI: https://doi.org/10.1021/es501923z
Lin, Y.L., Wei, Y., Sun, Y.H. (2012). Room-temperature synthesis and photocatalytic properties of lepidocrocite by monowavelength visible light irradiation. Journal of Molecular Catalysis A: Chemical, 353–354, 67–73. https://doi.org/10.1016/j.molcata.2011.11.006 DOI: https://doi.org/10.1016/j.molcata.2011.11.006
Tiar, K., Soualah, A., Bisio, C., Guidotti, M. (2024). Effect of initial solution pH on 4-nitrophenol oxidation through homogeneous/heterogeneous photo-Fenton process using goethite/H2O2 system. Journal of Photochemistry and Photobiology A: Chemistry, 447, 115184. https://doi.org/10.1016/j.jphotochem.2023.115184 DOI: https://doi.org/10.1016/j.jphotochem.2023.115184
Li, X., Niu, A., Yang, S. (2024). The reduction of nitrobenzene by Fe(II)-goethite-hematite heterogeneous systems: Insight from thermodynamic parameters of reduction potential. Journal of Environmental Management, 370, 122404. https://doi.org/10.1016/j.jenvman.2024.122404 DOI: https://doi.org/10.1016/j.jenvman.2024.122404
Liu, G., Yu, H., Zhou, T. (2022). Activation of peroxydisulfate by biogenic nanocomposites of reduced graphene oxide and goethite for non-radical selective oxidation of organic contaminants: Production of singlet oxygen and direct electron transfer. Chemical Engineering Journal, 430, 133177. https://doi.org/10.1016/j.cej.2021.133177 DOI: https://doi.org/10.1016/j.cej.2021.133177
Montes-Hernandez, G., Beck, P., & Findling, N. (2011). Fast precipitation of acicular goethite from ferric hydroxide gel under moderate temperature (30 and 70 C degrees). Crystal Growth & Design, 11 (6), 112264–2272. https://doi.org/10.1021/cg1016802 DOI: https://doi.org/10.1021/cg1016802
Gilbert, F., Refait, P., C., & Conforto, E. (2008). Synthesis of goethite from Fe(OH)2 precipitates: Influence of Fe(II) concentration and stirring speed. Journal of Physics and Chemistry of Solids, 69 (8), 2124-2130. https://doi.org/10.1016/j.jpcs.2008.03.010 DOI: https://doi.org/10.1016/j.jpcs.2008.03.010
Oulego, P., Villa-García, M.A., Lacaa, A., Diaz, M. (2016). Effect of the synthetic route on the structural, textural, morphological and catalytic properties of Iron (III) oxides and oxyhydroxides. Dalton Transactions, 45, 9446–9459. 10.1039/C6DT00247A DOI: https://doi.org/10.1039/C6DT00247A
Kuncser, A.C., Vlaicu, I.D., Pavel, O.D., Olar, R. (2021). Soft synthesis and characterization of goethite-based nanocomposites as promising cyclooctene oxidation catalysts. RSC Advances, 11, 27589-27602. https://doi.org/10.1039/D1RA04211D DOI: https://doi.org/10.1039/D1RA04211D
Cudennec, Y., Lecerf, A. (2006). The transformation of ferrihydrite into goethite or hematite, revisited. Journal of Solid State Chemistry, 179 (3), 716–722. https://doi.org/10.1016/j.jssc.2005.11.030 DOI: https://doi.org/10.1016/j.jssc.2005.11.030
Alves, C.A.P., Palharim, P.H., & Ramos, B. (2025). Photocatalytic ammonia synthesis from nitrogen in water using iron oxides: Comparative efficiency of goethite, magnetite, and hematite. Journal of Photochemistry and Photobiology A: Chemistry, 460, 116159. https://doi.org/10.1016/j.jphotochem.2024.116159 DOI: https://doi.org/10.1016/j.jphotochem.2024.116159
Adeoye A. O., Quadri, R. O., Lawal, O. S. (2023). Wet synthesis, characterization of goethite nanoparticles and its application in catalytic pyrolysis of palm kernel shell in TGA. Results in Surfaces and Interfaces, 11, 100118. https://doi.org/10.1016/j.rsurfi.2023.100118 DOI: https://doi.org/10.1016/j.rsurfi.2023.100118
Kosmulski, M., Maczka, E., Jartych, E., Rosenholm, J. B. (2003). Synthesis and characterization of goethite and goethite–hematite composite: experimental study and literature survey. Advances in Colloid and Interface Science, 103 (1), 57–76. https://doi.org/10.1016/S0001-8686(02)00083-0 DOI: https://doi.org/10.1016/S0001-8686(02)00083-0
Hinrichs, S., Grossmann, L., & Hankiewicz, B. (2020). Goethite Nanorods: Synthesis and Investigation of the Size Effect on Their Orientation within a Magnetic Field by SAXS. Nanomaterials, 10 (12), 2526. https://doi.org/10.3390/nano10122526 DOI: https://doi.org/10.3390/nano10122526
Mohapatra M., Gupta S., & Mishra B.K. (2010). pH and temperature dependent facile precipitation of nano-goethite particles in Fe(NO3)3–NaOH–NH3NH2HSO4–H2O medium. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 355 (1-3), 53–60. https://doi.org/10.1016/j.colsurfa.2009.11.029 DOI: https://doi.org/10.1016/j.colsurfa.2009.11.029
Meret, A., Ralf, K., & Ruben, K. (2019). Electrochemical Analysis of Changes in Iron Oxide Reducibility during Abiotic Ferrihydrite Transformation into Goethite and Magnetite. Environmental Science & Technology, 53 (7), 3568–3578. https://doi.org/10.1021/acs.est.8b07190 DOI: https://doi.org/10.1021/acs.est.8b07190
Mira, R., Ivana, O., Svetozar, M. (2013). The synthesis and microstructure of goethite particles precipitated in highly alkaline media. Journal of Alloys and Compounds, 559, 49–56. https://doi.org/10.1016/j.jallcom.2013.01.027 DOI: https://doi.org/10.1016/j.jallcom.2013.01.027
Xu, Y., Wang, H., Ye, S., et al. (2024). Goethite adaptation prompts alterations in antibiotic susceptibility and suppresses development of antibiotic resistance in bacteria. Science of The Total Environment, 916, 170248. https://doi.org/10.1016/j.scitotenv.2024.170248 DOI: https://doi.org/10.1016/j.scitotenv.2024.170248
Agresti, F., Isopi, J., Scattareggia Marchese, S., et al. (2025). Electrorheology of urea-functionalized lathlike goethite particles in silicone oil. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 704, 135509. https://doi.org/10.1016/j.colsurfa.2024.135509 DOI: https://doi.org/10.1016/j.colsurfa.2024.135509
Lyane, M.D., Gustavo, R.G., Miguel, A.S. Jr. (2022). Synthesis of nanostructured iron oxides and study of the thermal crystallization process using DSC and in situ XRD experiments. Materials Chemistry and Physics, 285, 126065. https://doi.org/10.1016/j.matchemphys.2022.126065 DOI: https://doi.org/10.1016/j.matchemphys.2022.126065
Zhang, Y-X., Jia, Y. (2014). A facile solution approach for the synthesis of akaganéite (β-FeOOH) nanorods and their ion-exchange mechanism toward As (V) ions. Applied Surface Science, 290, 102-106. https://doi.org/10.1016/j.apsusc.2013.11.007 DOI: https://doi.org/10.1016/j.apsusc.2013.11.007
Jia, Y., Luo, T., & Huang, X.J. (2013). Facile one-pot synthesis of lepidocrocite (γ-FeOOH) nanoflakes for water treatment. New Journal of Chemistry, 37 (8), 2551–2556. doi: http://dx.doi.org/10.1039/c3nj00509g DOI: https://doi.org/10.1039/c3nj00509g
Fang F., Jia Y., Wu P-Y., & Peng D-Y. (2015). Facile one-pot preparation of goethite/parabutlerite nanocomposites and their removal properties and mechanism toward As(V) ions. Applied Surface Science Journal, 324, 355–362. https://doi.org/10.1016/j.apsusc.2014.10.174 DOI: https://doi.org/10.1016/j.apsusc.2014.10.174
Ma, J., Zhang, X., He, Y., et al. (2015). Non-superparamagnetic iron-oxide architectures with controlled T2 contrast ability in magnetic resonance imaging. Applied Physics Letters, 107, 073701. https://doi.org/10.1063/1.4928914 DOI: https://doi.org/10.1063/1.4928914
Kang, S., Kim, G., Cheon, J. (2025). Corrugated-Surface Goethite Nanoparticles with Reduced Magnetization and Increased Surface Area for Enhanced T1 MRI Contrast Effect. ACS Applied Materials & Interfaces, 17(49), 66450–66459. https://doi.org/10.1021/acsami.5c18713 DOI: https://doi.org/10.1021/acsami.5c18713
Serup, J., Alsing, K.K., Olsen, O., et al. (2023). On the mechanism of painful burn sensation in tattoos on magnetic resonance imaging (MRI). Magnetic substances in tattoo inks used for permanent makeup (PMU) identified: Magnetite, goethite, and hematite. Skin Research and Technology, 29(3), e13281. https://doi.org/10.1111/srt.13281 DOI: https://doi.org/10.1111/srt.13281
Gulzar, A., Ayoub, N., Mir, J.F., et al. (2022). In vitro and in vivo MRI imaging and photothermal therapeutic properties of Hematite (α-Fe2O3) Nanorods. Journal of Materials Science: Materials in Medicine, 33 (10). https://doi.org/10.1007/s10856-021-06636-1 DOI: https://doi.org/10.1007/s10856-021-06636-1
Lunin, A.V., Kolychev, E.L., & Nikitin, M.P. (2019). Synthesis of highly-specific stable nanocrystalline goethite-like hydrous ferric oxide nanoparticles for biomedical applications by simple precipitation method. Journal of Colloid and Interface Science, 541, 143–149. https://doi.org/10.1016/j.jcis.2019.01.065 DOI: https://doi.org/10.1016/j.jcis.2019.01.065
Balzar, D., Audebrand, N., Daymond, M. R., Toby, B. H. (2004). Size-strain line-broadening analysis of ceria round-robin sample. Journal of Applied Crystallography, 37, 911–924. https://doi.org/10.1107/S0021889804022551 DOI: https://doi.org/10.1107/S0021889804022551
Nikolaeva, O.V., Terekhov, V.A. (2017). Improvement of laboratory phytotest for the ecological evaluation of soils. Eurasian Soil Science, 50, 1105-1114. 10.1134/S1064229317090058 DOI: https://doi.org/10.1134/S1064229317090058
Skalny, M., Rokowska, A., & Bajda, T. (2024). Nanoscale surface defects of goethite governing DNA adsorption process and formation of the Goethite-DNA conjugates, Chemosphere, 362, 142602. https://doi.org/10.1016/j.chemosphere.2024.142602 DOI: https://doi.org/10.1016/j.chemosphere.2024.142602
Gomez, M.A., Jiang, R., & Jia, Y. (2020). Further insights into the Fe(II) reduction of 2-line ferrihydrite: a semi in situ and in situ TEM study. Nanoscale Advances, 2, 4938-4950. https://doi.org/10.1039/d0na00643b DOI: https://doi.org/10.1039/D0NA00643B
Smith, S.J., Page, K., & Woodfield, B.F. (2012). Novel synthesis and structural analysis of ferrihydrite. Inorganic Chemistry, 51 (11), 6421–6424. https://doi.org/10.1021/ic300937f DOI: https://doi.org/10.1021/ic300937f
Su, R., Li, C., & Lan, S. (2024). Catalytic oxidation of Mn(II) on ferrihydrite and goethite surfaces and the subsequent oxidation and immobilization of coexisting Cr(III). Applied Geochemistry, 175, 106195. https://doi.org/10.1016/j.apgeochem.2024.106195 DOI: https://doi.org/10.1016/j.apgeochem.2024.106195
Okada, S., Takagi, K., Ozaki, K. (2016). Synthesis of submicron-sized acicular goethite and platelet-like hematite particles and dependence of magnetic properties of α-Fe particles on their shape and size. Materials Chemistry and Physics, 171, 171–177. https://doi.org/10.1016/j.matchemphys.2016.01.002 DOI: https://doi.org/10.1016/j.matchemphys.2016.01.002
Polyakov, A.Y., Sorkina, T.A., & Goodilin, E.A. (2013). Mössbauer spectroscopy of frozen solutions as a stepwise control tool in preparation of biocompatible humic-stabilized feroxyhyte nanoparticles. Hyperfine Interactions, 219, 113–120. https://doi.org/10.1007/s10751-013-0812-y DOI: https://doi.org/10.1007/s10751-013-0812-y
Pankratov, D.A., Dolzhenko, V.D., & Severin, A.V. (2017). Properties of iron-containing nanohydroxyapatite-based composites. Inorganic Materials, 53 (1), 115–124. https://doi.org/10.1134/S0020168517010125 DOI: https://doi.org/10.1134/S0020168517010125
Kulikova N.A., Polyakov A.Y., & Perminova I.V. (2017). Key roles of size and crystallinity of nanosized iron hydr (oxides) stabilized by humic substances in iron bioavailability to plants. Journal of Agricultural and Food Chemistry, 65 (51), 11157–11169. https://doi.org/10.1021/acs.jafc.7b03955 DOI: https://doi.org/10.1021/acs.jafc.7b03955
Yurkov, G.Y., Shashkeev, K.A., & Koksharov, Y.A. (2016). Synthesis and magnetic properties of cobalt ferrite nanoparticles in polycarbosilane ceramic matrix. Journal of Alloys and Compounds, 686, 421–430. https://doi.org/10.1016/j.jallcom.2016.06.025 DOI: https://doi.org/10.1016/j.jallcom.2016.06.025
Pankratov, D.A., Anuchina, M.M., Spiridonov, F.M., Krivtsov, G.G. (2020). Fe3–δO4 Nanoparticles Synthesized in the Presence of Natural Polyelectrolytes. Crystallography Reports, 65 (3), 393–397. https://doi.org/10.1134/S1063774520030244 DOI: https://doi.org/10.1134/S1063774520030244
Chernavskiy, P.A., Novakova, A.A., & Petrovskaya, G.A. (2023). Synthesis and Characterization of Hematite, Magnetite and Maghemite Supported on Silica Gel. Magnetochemistry, 9 (11), 228. https://doi.org/10.3390/magnetochemistry9110228 DOI: https://doi.org/10.3390/magnetochemistry9110228
Jones, D.H., Srivastava, K.K.P. (1986). Many-state relaxation model for the Mössbauer spectra of superparamagnets. Physical Review B, 34 (11), 7542–7548. https://doi.org/10.1103/PhysRevB.34.7542 DOI: https://doi.org/10.1103/PhysRevB.34.7542
Zharkynbaeva, R., Dzeranov, A., & Kydralieva, K. (2024). Exploring the synergistic effects of goethite intercalated coal in the presence of humic acids for enhanced growth of Sinapis alba. Chemical and Biological Technologies in Agriculture, 14, 1354. https://doi.org/10.1186/s40538-023-00530-4 DOI: https://doi.org/10.1186/s40538-023-00530-4
Pankratov, D.A., Dovletyarova, E.A., & Neaman, A. (2024). Deciphering the corrosion puzzle: Nano-iron-biochar composite — Not a quick fix for metal immobilization in peat soils. Applied Geochemistry, 166, 105982. https://doi.org/10.1016/j.apgeochem.2024.105982 DOI: https://doi.org/10.1016/j.apgeochem.2024.105982
Shoppert, A., Valeev, D., & Pankratov, D. (2022). High-Iron Bauxite Residue (Red Mud) Valorization Using Hydrochemical Conversion of Goethite to Magnetite. Materials, 15 (23), 8423. https://doi.org/10.3390/ma15238423 DOI: https://doi.org/10.3390/ma15238423
Brok, E., Frandsen, C., & Berhe, A. A. (2014). Magnetic properties of ultra-small goethite nanoparticles. Journal of Applied Physics D: Applied Physics, 47 (36), 365003. 10.1088/0022-3727/47/36/365003 DOI: https://doi.org/10.1088/0022-3727/47/36/365003
Shoppert, A., Valeev, D., Loginova, I., Pankratov, D. (2023). Low-Temperature Treatment of Boehmitic Bauxite Using the Bayer Reductive Method with the Formation of High-Iron Magnetite Concentrate. Materials, 16 (13), 4678. https://doi.org/10.3390/ma16134678 DOI: https://doi.org/10.3390/ma16134678
Polyakov, A.Y., Goldt, A.E., & Tretyakov, Y.D. (2012). Constrained growth of anisotropic magnetic δ-FeOOH nanoparticles in the presence of humic substances. CrystEngComm, 14 (23), 8097. https://doi.org/10.1039/C2CE25886B DOI: https://doi.org/10.1039/c2ce25886b
Dzeranov, A., Bondarenko, L., & Kydralieva, K. (2023). Iron Oxides Nanoparticles as Components of Ferroptosis-Inducing Systems: Screening of Potential Candidates. Magnetochemistry, 9 (1), 3. https://doi.org/10.3390/magnetochemistry9010003 DOI: https://doi.org/10.3390/magnetochemistry9010003
Rostovshchikova, T.N., Korobov, M.S., & Gubin, S.P. (2005). Catalytic conversions of chloroolefins over iron oxide nanoparticles 2. Isomerization of dichlorobutenes over iron oxide nanoparticles stabilized on the surface of ultradispersed poly(tetrafluoroethylene). Russian Chemical Bulletin, 54 (6), 1425–1432. https://doi.org/10.1007/s11172-005-0422-1 DOI: https://doi.org/10.1007/s11172-005-0422-1
Klygach, D.S., Vakhitov, M.G., & Trukhanov, A.V. (2021). MCC: Specific of preparation, correlation of the phase composition and electrodynamic properties. Journal of Magnetism and Magnetic Materials. 526, 167694. https://doi.org/10.1016/j.jmmm.2020.167694 DOI: https://doi.org/10.1016/j.jmmm.2020.167694
Campo, B.C., Rosseler, O., & Volp M.A. (2008). On the nature of goethite, Mn-goethite and Co-goethite as supports for gold nanoparticles Mater. Materials Chemistry and Physics, 109, 448-454. https://doi.org/10.1016/j.matchemphys.2007.12.014 DOI: https://doi.org/10.1016/j.matchemphys.2007.12.014
Tartaj, P., Amarilla, J.M. (2011). Iron oxide porous nanorods with different textural properties and surface composition: Preparation, characterization and electrochemical lithium storage capabilities. Journal of Power Sources, 196 (4), 2164–2170. https://doi.org/10.1016/j.jpowsour.2010.09.116 DOI: https://doi.org/10.1016/j.jpowsour.2010.09.116
Maiti, D., Aravindan, V., Madhavi, S., Sujatha, P. D. (2015). Electrochemical performance of hematite nanoparticles derived from spherical maghemite and elongated goethite particles. Journal of Power Sources, 276, 291–298. https://doi.org/10.1016/j.jpowsour.2014.11.097 DOI: https://doi.org/10.1016/j.jpowsour.2014.11.097
Harris, R.A. (2024). Phase transformation of magnetite and goethite nanoparticles controlled by pH: Experimental and simulation study of cuboid magnetic nanoparticles prepared with NaOH. Solid State Sciences, 148, 107416. https://doi.org/10.1016/j.solidstatesciences.2023.107416 DOI: https://doi.org/10.1016/j.solidstatesciences.2023.107416
Zhu, M., Wang, Y., Meng, D., Qin, X., Diao G. (2012). Hydrothermal Synthesis of Hematite Nanoparticles and Their Electrochemical Properties. The Journal of Physical Chemistry C, 116 (30), 16276–16285. https://doi.org/10.1021/jp304041m DOI: https://doi.org/10.1021/jp304041m
Chen, S.A., Heaney, P.J., & Stubbs, J.E. (2022). Hematite-goethite ratios at pH 2–13 and 25–170 ◦C: A time-resolved synchrotron X-ray diffraction study. Chemical Geology, 606, 120995. https://doi.org/10.1016/j.chemgeo.2022.120995 DOI: https://doi.org/10.1016/j.chemgeo.2022.120995
Akinwekomi, V., Maree, J.P., & Chatzisymeon, E. (2020). Beneficiation of acid mine drainage (AMD): A viable option for the synthesis of goethite, hematite, magnetite, and gypsum — Gearing towards a circular economy concept. Minerals Engineering, 148, 106204. https://doi.org/10.1016/j.mineng.2020.106204 DOI: https://doi.org/10.1016/j.mineng.2020.106204
Combes, J.M., Manceau, A., Calas, G. (1990). Formation of ferric oxides from aqueous solutions: A polyhedral approach by X-ray Absorption Spectroscopy: II. Hematite formation from ferric gels. Geochimica et Cosmochimica Acta, 54, 1083–1091. https://doi.org/10.1016/0016-7037(90)90440-V DOI: https://doi.org/10.1016/0016-7037(90)90440-V
Vu, H.P., Shaw, S., Brinza, L., Benning, L.G. (2013). Partitioning of Pb(II) during goethite and hematite crystallization: Implications for Pb transport in natural systems. Applied Geochemistry, 39, 119-128. https://doi.org/10.1016/j.apgeochem.2013.10.001 DOI: https://doi.org/10.1016/j.apgeochem.2013.10.001
De La Vega, J.C., Elischer, P., Schneider, T., Häfeli, U.O. (2013). Uniform polymer microspheres: monodispersity criteria, methods of formation and applications. Nanomedicine, 8, 265–285. https://doi.org/10.2217/nnm.12.210 DOI: https://doi.org/10.2217/nnm.12.210
Chen, K., Guo, C., & Dang Z. (2024). Using machine learning to explore oxyanion adsorption ability of goethite with different specific surface area. Environmental Pollution, 343, 123162. https://doi.org/10.1016/j.envpol.2023.123162 DOI: https://doi.org/10.1016/j.envpol.2023.123162
Belloni, C., Korving, L., & Dugulan, A.I. (2024). Zn induced surface modification of stable goethite nanoparticles for improved regenerative phosphate adsorption. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 687, 133476. https://doi.org/10.1016/j.colsurfa.2024.133476 DOI: https://doi.org/10.1016/j.colsurfa.2024.133476
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Sofia M. Mulyukina, Artur A. Dzeranov, Denis A. Pankratov, Lyubov S. Bondarenko, Gulsara D. Kugabaeva, Mikhail V. Prokof`ev, Kamila A. Kydralieva

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



