Dispersion Technique May Change Structure and Bio-Oxidative Activity of Magnetic MOF Nanoparticles
DOI:
https://doi.org/10.31489/2959-0663/1-26-8Keywords:
Metal organic framework, Magnetic iron oxide nanoparticles, Nanosuspensions, Sample preparation operations, Grinding in the mortar, Ultrasonication, Structure, Oxidative properties, MOFAbstract
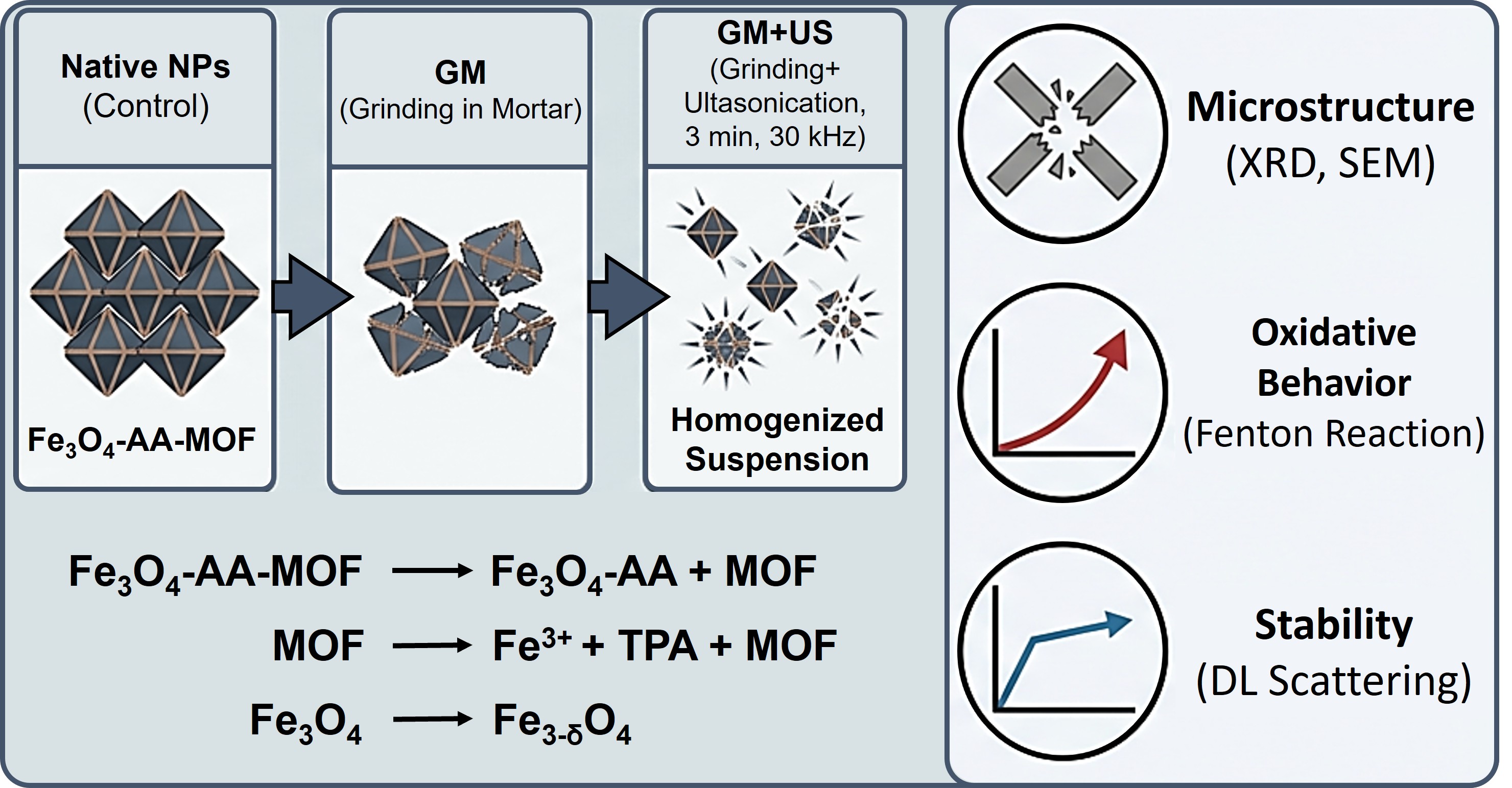
The overlooked influence of routine operations on hydrophobic nanosuspensions can induce polymorphic transformation, a critical factor that contributes to their instability and significantly limits their applications. Techniques such as ultrasonication are commercially employed for producing nanosuspensions. However, the impact of routine operations, including mechanical grinding and ultrasound, on obtaining stable suspensions has not received sufficient attention, despite their potential to alter microstructure, morphology, and consequently, functional properties. In this study, Fe3O4-asсorbic acid/ metal-organic coordination polymer (MOF) MIL-88b (Fe3O4-AA-MOF) nanoparticles (NPs) were subjected to grinding in a mortar (GM). Subsequently, 1.0 wt.% of aqueous suspensions were ultrasonicated (GM+US) for 3 min at 30 kHz and 37 °C. The structure and oxidative properties of the homogenized suspensions were investigated using X-ray diffraction technique, dynamic light scattering (DLS), and scanning electronic microscope (SEM), with native NPs serving as a control. Homogenization treatment significantly affected the microstructure and oxidative behavior of Fe3O4-AA-MOF NPs. The combination of milling and ultrasound led to a change in the stoichiometry of magnetite, partial destruction of MOF, and simultaneously, an acceleration of the Fenton reaction and increased stability of NPs in suspension. These findings underscore that the influence of routine sample preparation operations on the functional properties of NPs cannot be underestimated.
References
Cherepanova, V.A., Gordeev, E.G., & Ananikov, V.P. (2025). Magnetic Stirring May Cause Irreproducible Results in Chemical Reactions. JACS Au, 5(8), 3789–3798. https://doi.org/10.1021/jacsau.5c00412 DOI: https://doi.org/10.1021/jacsau.5c00412
Mirkin, C.A., Petrosko, S.H., Artzi, N., Aydin, K., Biaggne, A., Brinker, C.J., Bujold, K.E., Cao, Y.C., Chan, R.R., Chen, C., Chen, P.C., Chen, X., Chevalier, O.J.G.L., Choi, C.H.J., Crooks, R.M., Dravid, V.P., Du, J.S., Ebrahimi, S.B., Fan, H., Farha, O.K., Figg, C.A., Fink, T.D., Forsyth, C.M., Fuchs, H., Geiger, F.M., Gianneschi, N.C., Gibson, K.J., Ginger, D.S., Guo, S.S., Hanes, J.S., Hao, L., Huang, J., Hunter, B.M., Huo, F., Hwang, J., Jin, R., Kelley, S.O., Kempa, T.J., Kim, Y., Kudruk, S., Kumari, S., Landy, K.M., Lee, K.B., Leon, N.J., Li, J., Li, Y., Li, Z., Liu, B., Liu, G., Liu, X., Liz-Marzán, L.M., Lorch, J.H., Luo, T., Macfarlane, R.J., Millstone, J.E., Mrksich, M., Murphy, C.J., Naik, R.R., Nel, A.E., Oetheimer, C., Hedlund Orbeck, J.K., Park, S.J., Partridge, B.E., Peppas, N.A., Personick, M.L., Raj, A., Ramani, N., Ross, M.B., Ross, S.B., Sargent, E.H., Sengupta, T., Schatz, G.C., Seferos, D.S., Seideman, T., Seo, S.E., Shen, B., Shim, W., Shin, D., Simon, U., Sinegra, A.J., Smith, P.T., Spokoyny, A.M., Stang, P.J., Stegh, A.H., Stoddart, J.F., Swearer, D.F., Tan, W., Teplensky, M.H., Thaxton, C.S., Walt, D.R., Wang, M.X., Wang, Z., Wei, W.D., Weiss, P.S., Winegar, P.H., Xia, Y., Xie, Y., Xu, X., Yang, P., Yang, Y., Ye, Z., Yoon, K.R., Zhang, C., Zhang, H., Zhang, K., Zhang, L., Zhang, X., Zhang, Y., Zheng, Z., Zhou, W., Zhu, S. & Zhu, W. (2025). 33 Unresolved Questions in Nanoscience and Nanotechnology. ACS Nano, 19, 36, 31933–31968. https://doi.org/10.1021/acsnano.5c12854 DOI: https://doi.org/10.1021/acsnano.5c12854
Babayevska, N., Przysiecka, L., Iatsunskyi, I., Nowaczyk, G., Jarek, M., Janiszewska, E., & Jurga, S. (2023). Author Correction: ZnO size and shape effect on antibacterial activity and cytotoxicity profile. Scientific reports, 13(1), 12465. https://doi.org/10.1038/s41598-023-39615-3 DOI: https://doi.org/10.1038/s41598-023-39615-3
Zhang, B., Sai Lung, P., Zhao, S., Chu, Z., Chrzanowski, W., & Li, Q. (2017). Shape dependent cytotoxicity of PLGA-PEG nanoparticles on human cells. Scientific reports, 7(1), 7315. https://doi.org/10.1038/s41598-017-07588-9 DOI: https://doi.org/10.1038/s41598-017-07588-9
Subhan, M.A., Yalamarty, S.S.K., Filipczak, N., Parveen, F., & Torchilin, V.P. (2021). Recent Advances in Tumor Targeting via EPR Effect for Cancer Treatment. Journal of personalized medicine, 11(6), 571. https://doi.org/10.3390/jpm11060571 DOI: https://doi.org/10.3390/jpm11060571
Rizvi, S.A.A., & Saleh, A.M. (2018). Applications of nanoparticle systems in drug delivery technology. Saudi pharmaceutical journal: SPJ: the official publication of the Saudi Pharmaceutical Society, 26(1), 64–70. https://doi.org/10.1016/j.jsps.2017.10.012 DOI: https://doi.org/10.1016/j.jsps.2017.10.012
Öztürk, K., Kaplan, M., & Çalış, S. (2024). Effects of nanoparticle size, shape, and zeta potential on drug delivery. International journal of pharmaceutics, 666, 124799. https://doi.org/10.1016/j.ijpharm.2024.124799 DOI: https://doi.org/10.1016/j.ijpharm.2024.124799
Bhatia, S. (2016). Natural Polymer Drug Delivery Systems. Springer International Publishing. https://doi.org/10.1007/978-3-319-41129-3 DOI: https://doi.org/10.1007/978-3-319-41129-3
Bhardwaj, V., Kaushik, A., Khatib, Z.M., Nair, M., & McGoron, A.J. (2019). Recalcitrant Issues and New Frontiers in Nano-Pharmacology. Frontiers in pharmacology, 10, 1369. https://doi.org/10.3389/fphar.2019.01369 DOI: https://doi.org/10.3389/fphar.2019.01369
Honary, S., & Zahir, F. (2013). Effect of Zeta Potential on the Properties of Nano-Drug Delivery Systems — A Review (Part 2). Tropical Journal of Pharmaceutical Research, 12(2), 265–273. https://doi.org/10.4314/tjpr.v12i2.20 DOI: https://doi.org/10.4314/tjpr.v12i2.20
Ding, S., Khan, A. I., Cai, X., Song, Y., Lyu, Z., Du, D., Dutta, P., & Lin, Y. (2020). Overcoming blood-brain barrier transport: Advances in nanoparticle-based drug delivery strategies. Materials today (Kidlington, England), 37, 112–125. https://doi.org/10.1016/j.mattod.2020.02.001 DOI: https://doi.org/10.1016/j.mattod.2020.02.001
Clogston, J.D., & Patri, A.K. (2011). Zeta potential measurement. Methods in molecular biology (Clifton, N.J.), 697, 63–70. https://doi.org/10.1007/978-1-60327-198-1_6 DOI: https://doi.org/10.1007/978-1-60327-198-1_6
Mahbubul, I.M., Saidur, R., Amalina, M.A., Elcioglu, E.B., & Okutucu-Ozyurt, T. (2015). Effective ultrasonication process for better colloidal dispersion of nanofluid. Ultrasonics sonochemistry, 26, 361–369. https://doi.org/10.1016/j.ultsonch.2015.01.005 DOI: https://doi.org/10.1016/j.ultsonch.2015.01.005
Müller, R.H., Jacobs, C., & Kayser, O. (2001). Nanosuspensions as particulate drug formulations in therapy. Rationale for development and what we can expect for the future. Advanced drug delivery reviews, 47(1), 3–19. https://doi.org/10.1016/s0169-409x(00)00118-6 DOI: https://doi.org/10.1016/S0169-409X(00)00118-6
Hwang, Y., Lee, J., Lee, J., Jeong, Y., Cheong, S., Ahn, Y.C., & Kim, S.H. (2008). Production and dispersion stability of nanoparticles in nanofluids. Powder Technology, 186, 145–153. https://doi.org/10.1016/j.powtec.2007.11.020 DOI: https://doi.org/10.1016/j.powtec.2007.11.020
Kamala Kumari, P.V. & Srinivasa Rao, Y. (2017). Nanosuspensions: A review. International Journal of Pharmacy. 7(2), 77–89.
Taurozzi, J., Hackley, V. & Wiesner, M. (2012). Preparation of Nanoparticle Dispersions from Powdered Material Using Ultrasonic Disruption — Version 1.1. National Institute of Standards and Technology, Gaithersburg, MD. https://doi.org/10.6028/NIST.SP.1200-2 DOI: https://doi.org/10.6028/NIST.SP.1200-2
Sobenin, D.V., Solovov, R.D. & Ershov, B.G. (2026). Transformation of zerovalent iron nanoparticles in isopropanol into oxide particles: effect of water and air on mechanism and kinetics. Journal of Molecular Liquids. 447, 129333. https://doi.org/10.1016/j.molliq.2026.129333 DOI: https://doi.org/10.1016/j.molliq.2026.129333
Taurozzi, J.S., Hackley, V.A., & Wiesner, M.R. (2011). Ultrasonic dispersion of nanoparticles for environmental, health and safety assessment—issues and recommendations. Nanotoxicology, 5(4), 711–729. https://doi.org/10.3109/17435390.2010.528846 DOI: https://doi.org/10.3109/17435390.2010.528846
Mane, N.P., Rane, B.R. & Jain, A.S. (2024). Advances in Nanosuspension Technology: Current Trends and Future Horizons. Nano Biomedicine and Engineering, 16(4): 574–587. https://doi.org/10.26599/NBE.2024.9290092 DOI: https://doi.org/10.26599/NBE.2024.9290092
Jacob, S., Nair, A. B., & Shah, J. (2020). Emerging role of nanosuspensions in drug delivery systems. Biomaterials research, 24, 3. https://doi.org/10.1186/s40824-020-0184-8 DOI: https://doi.org/10.1186/s40824-020-0184-8
Li, H., Zhu, M., Pang, Y., Du, H. & Liu, T. (2016). Influences of ultrasonic irradiation on the morphology and structure of nanoporous Co nanoparticles during chemical dealloying. Progress in Natural Science: Materials International. 26, 562–566. https://doi.org/10.1016/j.pnsc.2016.12.002 DOI: https://doi.org/10.1016/j.pnsc.2016.12.002
Zhang, X., Jiang, X., Qu, S., Zhang, H., Yang, W., Lu, W. (2022). Effects of ultrasonication on the microstructures and mechanical properties of carbon nanotube films and their based composites. Composites Science and Technology. 221, 109136. https://doi.org/10.1016/j.compscitech.2021.109136 DOI: https://doi.org/10.1016/j.compscitech.2021.109136
Li, Y. -H., Wang, C.-C., Yi, X.-H. & Chu, H.-Y. (2023). UiO-66(Zr)-based functional materials for water purification: An updated review. Environmental Functional Materials. 2(2): 93–132. https://doi.org/10.1016/j.efmat.2024.02.001 DOI: https://doi.org/10.1016/j.efmat.2024.02.001
Lin, Y., Min, K., Ma, W., Yang, X., Lu, D., Lin, Z., Liu Q. & Jiang, G. (2024). Probing the stability of metal–organic frameworks by structure-responsive mass spectrometry imaging. Chemical Science. 15, 3698–3706. https://doi.org/10.1039/D4SC00021H DOI: https://doi.org/10.1039/D4SC00021H
Bondarenko, L., Baimuratova, R., Dzeranov, A., Pankratov, D., Kicheeva, A., Sushko, E., Kudryasheva, N., Valeev, R., Tropskaya, N., Dzhardimalieva, G. & Kydralieva, K. (2024). Fenton reaction-driven pro-oxidant synergy of ascorbic acid and iron oxide nanoparticles in MIL-88B(Fe). New Journal of Chemistry. 48, 10142–10160. https://doi.org/10.1039/D4NJ00963K DOI: https://doi.org/10.1039/D4NJ00963K
Elmore, W.C. (1938). Ferromagnetic Colloid for Studying Magnetic Structures. Physical Review. 54, 309–310. https://doi.org/10.1103/PhysRev.54.309 DOI: https://doi.org/10.1103/PhysRev.54.309
Xu, X., Ma, J., Wang, A. & Zheng, N. (2023). N-Sulfonyl amidine polypeptides: new polymeric biomaterials with conformation transition responsive to tumor acidity. Chemical Science. 15(5), 1769–1781. https://doi.org/10.1039/d3sc05504c DOI: https://doi.org/10.1039/D3SC05504C
Sun, R., Wang, Y., Shi, W., Zhang, H., Liu, J. & He, W. (2024). Acidity-Triggered “Sticky Spotlight”: CCK2R-targeted TME-sensitive NIR fluorescent probes for tumor imaging in vivo. Bioconjugate Chemistry. 35(4), 528–539. https://doi.org/10.1021/acs.bioconjchem.4c00040 DOI: https://doi.org/10.1021/acs.bioconjchem.4c00040
Liu, Y., Huang, S., Sun, H.J., Liu, Y., Liang, L., Nan, Q., Wang, T., Chen, Z., Tang, J., Hu, C. & Zhao, J.R. (2023). Amorphous Cu-Mn hopcalite as novel Fenton-like catalyst for H2O2-activated degradation of tetracycline at circumneutral pH. Journal of Science: Advanced Materials and Devices. 8(3), 100603. https://doi.org/10.1016/j.jsamd.2023.100603 DOI: https://doi.org/10.1016/j.jsamd.2023.100603
Maniakova, G. & Rizzo, L. (2023). Pharmaceuticals degradation and pathogens inactivation in municipal wastewater: A comparison among UVC photo-Fenton with chelating agents, UVC/H2O2 and ozonation. Journal of Environmental Chemical Engineering. 11(6), 111356. https://doi.org/10.1016/j.jece.2023.111356 DOI: https://doi.org/10.1016/j.jece.2023.111356
Schwaminger, S.P., Syhr, C., & Berensmeier, S. (2020). Controlled Synthesis of Magnetic Iron Oxide Nanoparticles: Magnetite or Maghemite? Crystals, 10, 214. https://doi.org/10.3390/cryst10030214 DOI: https://doi.org/10.3390/cryst10030214
Gorski, C.A., & Scherer, M.M. (2010). Determination of nanoparticulate magnetite stoichiometry by Mössbauer spectroscopy, acidic dissolution, and powder X-ray diffraction: A critical review. American Mineralogist, 95(7), 1017–1026. https://doi.org/10.2138/am.2010.3435 DOI: https://doi.org/10.2138/am.2010.3435
Bogart, L.K., Blanco-Andujar, C. & Pankhurst, Q.A. (2018). Environmental oxidative aging of iron oxide nanoparticles. Applied Physics Letters, 113, 133701. https://doi.org/10.1063/1.5050217 DOI: https://doi.org/10.1063/1.5050217
Saber Braim, F., Noor Ashikin Nik Ab Razak, N., Abdul Aziz, A., Qasim Ismael, L., & Kayode Sodipo, B. (2022). Ultrasound assisted chitosan coated iron oxide nanoparticles: Influence of ultrasonic irradiation on the crystallinity, stability, toxicity and magnetization of the functionalized nanoparticles. Ultrasonics sonochemistry, 88, 106072. https://doi.org/10.1016/j.ultsonch.2022.106072 DOI: https://doi.org/10.1016/j.ultsonch.2022.106072
Stolyar, S.V., Bayukov, O.A., Yaroslavtsev, R.N., Knyazev, Yu.V., Ladygina, V.P., Gerasimova, Yu.V. & Iskhakov, R.S. (2019). Ion Reduction in Iron Oxide and Oxihydroxide Nanoparticles During Ultrasonic Treatment. Advanced Powder Technology. 30. 2620–2625. https://doi.org/10.1016/j.apt.2019.08.009 DOI: https://doi.org/10.1016/j.apt.2019.08.009
Kim, D., Lee, G., Oh, S. & Oh, M. (2019). Unbalanced MOF-on-MOF growth for the production of a lopsided core–shell of MIL-88B@MIL-88A with mismatched cell parameters. Chemical Communications. 55, 43–46. https://doi.org/10.1039/C8CC08456D DOI: https://doi.org/10.1039/C8CC08456D
Bondarenko, L., Baimuratova, R., Reindl, M., Zach, V., Dzeranov, A., Pankratov, D., Kydralieva, K., Dzhardimalieva, G., Kolb, D., Wagner, F.E., Schwaminger, S.P. (2024). Dramatic change in the properties of magnetite-modified MOF particles depending on the synthesis approach. Heliyon, 10, 6, e27640. https://doi.org/10.1016/j.heliyon.2024.e27640 DOI: https://doi.org/10.1016/j.heliyon.2024.e27640
Pukazhselvan, D., Granadeiro, C.M., Loureiro, F.J., Shaula, A.L., Mikhalev, S.M., Goncalves, G., & Fagg, D.P. (2023). Comparative analyses of MIL-88B (Fe) and MIL-100 (Fe) metal organic frameworks as active anode materials for Li ion batteries. Electrochimica Acta, 465, 142989. https://doi.org/10.1016/j.electacta.2023.142989 DOI: https://doi.org/10.1016/j.electacta.2023.142989
Ramos, M.D.N., Silva, G.L.S., Lessa, T.L. & Aguiar, A. (2022). Study of kinetic parameters related to dyes oxidation in ascorbic acid-mediated Fenton processes. Process Safety and Environmental Protection. 168, 1131–1141. https://doi.org/10.1016/j.psep.2022.10.083 DOI: https://doi.org/10.1016/j.psep.2022.10.083
Gogate, P. R., & Patil, P. N. (2015). Combined treatment technology based on synergism between hydrodynamic cavitation and advanced oxidation processes. Ultrasonics sonochemistry, 25, 60–69. https://doi.org/10.1016/j.ultsonch.2014.08.016 DOI: https://doi.org/10.1016/j.ultsonch.2014.08.016
Pouran, S.R., Aziz, A.R., & Daud, W.M. (2015). Review on the main advances in photo-Fenton oxidation system for recalcitrant wastewaters. Journal of Industrial and Engineering Chemistry, 21, 53–69. https://doi.org/10.1016/j.jiec.2014.05.005 DOI: https://doi.org/10.1016/j.jiec.2014.05.005
Li, J., Zhao, L., Qin, L., Tian, X., Wang, A., Zhou, Y. & Chen, Y. (2016). Removal of refractory organics in nanofiltration concentrates of municipal solid waste leachate treatment plants by combined Fenton oxidative-coagulation with photo–Fenton processes. Chemosphere, 146, 442–449. https://doi.org/10.1016/j.chemosphere.2015.12.069 DOI: https://doi.org/10.1016/j.chemosphere.2015.12.069
Pouran, S.R., Raman, A.A., & Daud, W.M. (2014). Review on the application of modified iron oxides as heterogeneous catalysts in Fenton reactions. Journal of Cleaner Production, 64, 24–35. https://doi.org/10.1016/j.jclepro.2013.09.013 DOI: https://doi.org/10.1016/j.jclepro.2013.09.013
Pouran, S.R., Aziz, A.A., Daud, W.M.A.W., & Embong, Z. (2015). Niobium substituted magnetite as a strong heterogeneous Fenton catalyst for wastewater treatment. Applied Surface Science, 351, 175–187. https://doi.org/10.1016/j.apsusc.2015.05.131 DOI: https://doi.org/10.1016/j.apsusc.2015.05.131
Costa, R.C., Lelis, M.F.F., Oliveira, L.C.A., Fabris, J.D., Ardisson, J.D., Rios, R.R.V.A., Silva, C.N. & Lago, R. M. (2006). Novel active heterogeneous Fenton system based on Fe3− xMxO4 (Fe, Co, Mn, Ni): The role of M2+ species on the reactivity towards H2O2 reactions. Journal of hazardous materials, 129(1-3), 171–178. https://doi.org/10.1016/j.jhazmat.2005.08.028 DOI: https://doi.org/10.1016/j.jhazmat.2005.08.028
Magalhães, F., Pereira, M.C., Botrel, S.E.C., Fabris, J.D., Macedo, W.A., Mendonça, R., Lago, R.M. & Oliveira, L.C.A. (2007). Cr-containing magnetites Fe3−xCrxO4: The role of Cr3+ and Fe2+ on the stability and reactivity towards H2O2 reactions. Applied Catalysis A: General. 332, 115–123. https://doi.org/10.1016/j.apcata.2007.08.002 DOI: https://doi.org/10.1016/j.apcata.2007.08.002
Liang, X., Zhong, Y., He, H., Yuan, P., Zhu, J., Zhu, S., & Jiang, Z. (2012). The application of chromium substituted magnetite as heterogeneous Fenton catalyst for the degradation of aqueous cationic and anionic dyes. Chemical Engineering Journal, 191, 177–184. https://doi.org/10.1016/j.cej.2012.03.001 DOI: https://doi.org/10.1016/j.cej.2012.03.001
Pouran, S.R., Bayrami, A., Aziz, A.A., Daud, W.M.A.W., & Shafeeyan, M.S. (2016). Ultrasound and UV assisted Fenton treatment of recalcitrant wastewaters using transition metal-substituted-magnetite nanoparticles. Journal of Molecular Liquids, 222, 1076–1084. https://doi.org/10.1016/j.molliq.2016.07.120 DOI: https://doi.org/10.1016/j.molliq.2016.07.120
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Lyubov S. Bondarenko, Artur A. Dzeranov, Nataliya S. Tropskaya, Kamila A. Kydralieva

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



