Size and Magnetization Control of Magnetite NPs via Ethylene Glycol and Temperature for Ferrofluid and Magnetotargeting: Model Experiments
DOI:
https://doi.org/10.31489/2959-0663/1-26-10Keywords:
Synthesis of magnetic nanoparticles, Ethylene glycol, Medium viscosity, Synthesis temperature, Oxidation, Size control, Saturation magnetization, Rheology, Magnetic captureAbstract
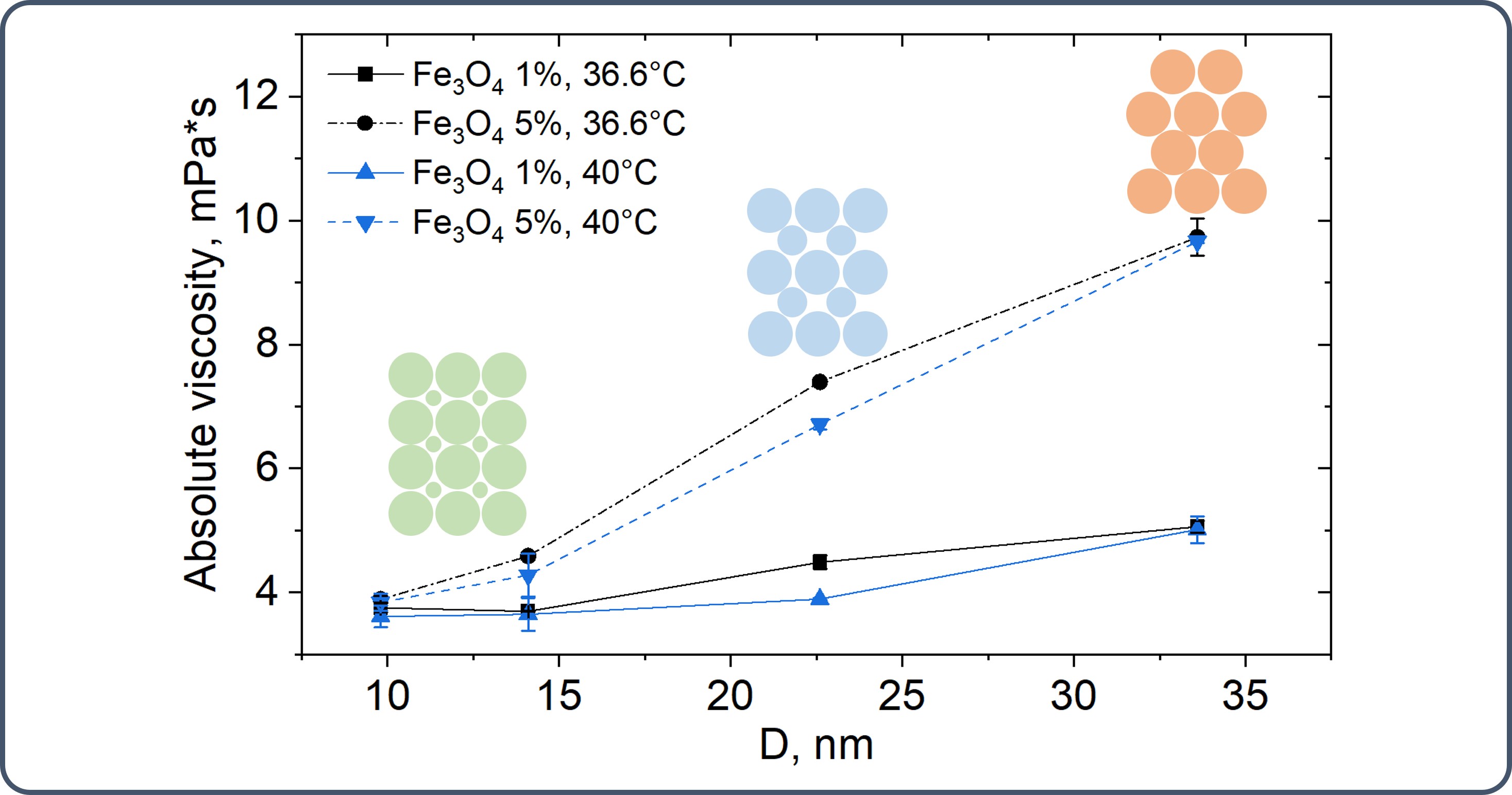
Magnetic nanoparticles (NPs) are highly promising materials for diverse biomedical applications, particularly in magnetotargeting, owing to their tunable magnetic properties. Achieving precise control over their synthesis parameters is critical for optimizing these properties and ensuring their efficacy. This study investigates the impact of synthesis the impact of synthesis conditions on the properties of magnetite (Fe3O4) nanoparticles. A set of magnetite NPs were prepared via co-precipitation of ferrous and ferric ions by a base in ambient conditions, systematically varying the ratio of ethylene glycol/water (EG/water) solvents and temperatures (4, 20, and 70 °C). Crystal structure, morphology and magnetic parameters were analyzed. Additionally, rheological experiments were conducted to study the dynamic viscosity of ferrofluids containing varying sizes and concentrations NPs in polyglucinum. Results showed that Fe3O4 NPs size linearly increased with the EG/water volume ratio only at 70 °C. Increasing EG from 0 % to 50 % elevated magnetite stoichiometry from Fe2.72O4 to Fe2.912O4 with higher concentrations reducing it. The effect of temperature on stoichiometry varied depending on the ethylene glycol (EG) content. At low EG content, stoichiometry decreased with increasing temperature. But, at higher EG content, maximum stoichiometry was observed at 20 °C. Increasing polydispersity of NPs (0.07–0.13) decreased viscosity in polyglucinum from ~10 to ~4 mPa s (1–5 % EG). Evaluation tests of the magnetic induction gradient on NPs capture in a flow-through setup were also performed. A sigmoidal relationship (R2 = 0.97) was established between the NPs capture efficiency and the magnetic induction gradient. These findings provide valuable insights for optimizing the synthesis and suspension performance for magnetic fluids for magnetotargeting applications.
References
Tian, Y., Yu, B., Li, X., & Li, K. (2011). Facile solvothermal synthesis of monodisperse Fe3O4 nanocrystals with precise size control of one nanometre as potential MRI contrast agents. Journal of Materials Chemistry, 21(8), 2476–2481. https://doi.org/10.1039/C0JM02913K DOI: https://doi.org/10.1039/c0jm02913k
Turrina, C., Oppelt, A., Mitzkus, M., Berensmeier, S., & Schwaminger, S. P. (2022). Silica-coated superparamagnetic iron oxide nanoparticles: New insights into the influence of coating thickness on the particle properties and lasioglossin binding. MRS Communications, 12(5), 632–639. https://doi.org/10.1557/s43579-022-00228-y DOI: https://doi.org/10.1557/s43579-022-00228-y
Ulbrich, K., Hola, K., Subr, V., Bakandritsos, A., Tucek, J., & Zboril, R. (2016). Targeted drug delivery with polymers and magnetic nanoparticles: covalent and noncovalent approaches, release control, and clinical studies. Chemical reviews, 116(9), 5338–5431. https://doi.org/10.1021/acs.chemrev.5b00589 DOI: https://doi.org/10.1021/acs.chemrev.5b00589
Reindl, M., Zach, V., & Schwaminger, S. P. (2025). Biocompatible Poly(acrylic acid-co-methacrylic acid)-Coated Iron Oxide Nanoparticles for Enhanced Adsorption and Antimicrobial Activity of Lasioglossin-III. ACS Applied Materials & Interfaces, 17(11), 16644-16657. https://doi.org/10.1021/acsami.4c22603 DOI: https://doi.org/10.1021/acsami.4c22603
Eigenfeld, M., Reindl, M., Sun, X., & Schwaminger, S. P. (2024). Exploring Multi-Parameter Effects on Iron Oxide Nanoparticle Synthesis by SAXS Analysis. Crystals, 14(11), 961. https://doi.org/10.3390/cryst14110961 DOI: https://doi.org/10.3390/cryst14110961
Pham, A. L. T., Lee, C., Doyle, F. M., & Sedlak, D. L. (2009). A silica-supported iron oxide catalyst capable of activating hydrogen peroxide at neutral pH values. Environmental science & technology, 43(23), 8930–8935. https://doi.org/10.1021/es902296k DOI: https://doi.org/10.1021/es902296k
Saman, D., Bondarenko, L. S., Baimuratova, R. K., Dzeranov, A. A., Dzhardimalieva, G. I., Tropskaya, N. S., & Kydralieva, K. A. (2024). A Statistical Design Approach for an Effective Catalyst in the Fenton Reaction in Case of Fe3O4-MOF MIL-88b (Fe) in Methylene Blue Degradation Kinetics. Eurasian Journal of Chemistry, 29(115), 16–29. https://doi.org/10.31489/2959-0663/3-24-15 DOI: https://doi.org/10.31489/2959-0663/3-24-15
Dzeranov, A., Pankratov, D., Bondarenko, L., Telegina, L., Dzhardimalieva, G., Saman, D., & Kydralieva, K. (2024). Humic acids-modified mesoporous silica encapsulating magnetite: crystal and surface characteristics. CrystEngComm, 26(24), 3250–3262. https://doi.org/10.1039/D4CE00281D DOI: https://doi.org/10.1039/D4CE00281D
Bondarenko, L., Baimuratova, R., Dzeranov, A., Pankratov, D., Kicheeva, A., Sushko, E., ... & Kydralieva, K. (2024). Fenton reaction-driven pro-oxidant synergy of ascorbic acid and iron oxide nanoparticles in MIL-88B (Fe). New Journal of Chemistry, 48(22), 10142–10160. https://doi.org/10.1039/D4NJ00963K DOI: https://doi.org/10.1039/D4NJ00963K
Bondarenko, L., Baimuratova, R., Reindl, M., Zach, V., Dzeranov, A., Pankratov, D., ... & Schwaminger, S. P. (2024). Designed magnetic nanoparticles for ferroptosis: Release of iron ions from metal-organic frameworks modified with iron oxides. Materials Today Chemistry, 42, 102332. https://doi.org/10.1016/j.mtchem.2024.102332 DOI: https://doi.org/10.1016/j.mtchem.2024.102332
Dzeranov, A., Bondarenko, L., Pankratov, D., Prokof‘ev, M., Dzhardimalieva, G., Jorobekova, S., ... & Kydralieva, K. (2022). Iron oxides nanoparticles as components of ferroptosis-inducing systems: screening of potential candidates. Magnetochemistry, 9(1), 3. https://doi.org/10.3390/magnetochemistry9010003 DOI: https://doi.org/10.3390/magnetochemistry9010003
Sahoo, Y., Goodarzi, A., Swihart, M. T., Ohulchanskyy, T. Y., Kaur, N., Furlani, E. P., & Prasad, P. N. (2005). Aqueous ferrofluid of magnetite nanoparticles: fluorescence labeling and magnetophoretic control. The Journal of Physical Chemistry B, 109(9), 3879–3885. https://doi.org/10.1021/jp045402y DOI: https://doi.org/10.1021/jp045402y
Lee, N., & Hyeon, T. (2012). Designed synthesis of uniformly sized iron oxide nanoparticles for efficient magnetic resonance imaging contrast agents. Chemical Society Reviews, 41(7), 2575–2589. https://doi.org/10.1039/C1CS15248C DOI: https://doi.org/10.1039/C1CS15248C
Jurgons, R., Seliger, C., Hilpert, A., Trahms, L., Odenbach, S., & Alexiou, C. (2006). Drug loaded magnetic nanoparticles for cancer therapy. Journal of Physics: Condensed Matter, 18(38), S2893. https://doi.org/10.1088/0953-8984/18/38/S24 DOI: https://doi.org/10.1088/0953-8984/18/38/S24
Pankhurst, Q. A., Connolly, J., Jones, S. K., & Dobson, J. J. J. (2003). Applications of magnetic nanoparticles in biomedicine. Journal of physics D: Applied physics, 36(13), R167. https://doi.org/10.1088/0022-3727/36/13/201 DOI: https://doi.org/10.1088/0022-3727/36/13/201
Laurent, S., Forge, D., Port, M., Roch, A., Robic, C., Vander Elst, L., & Muller, R. N. (2008). Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chemical reviews, 108(6), 2064–2110. https://doi.org/10.1021/cr068445e DOI: https://doi.org/10.1021/cr068445e
Spicher, M. T., Schwaminger, S. P., Von Der Haar-Leistl, D., Peralta, M. M., Mikacevic, G., Wagner, F. E., & Berensmeier, S. (2024). Pilot-scale co-precipitation synthesis of a novel active ingredient made of ultrasmall iron (oxyhydr) oxide nanoparticles for the treatment of hyperphosphatemia. RSC advances, 14(23), 16117–16127. https://doi.org/10.1039/D4RA02719A DOI: https://doi.org/10.1039/D4RA02719A
Qiao, L., Fu, Z., Li, J., Ghosen, J., Zeng, M., Stebbins, J., ... & Swihart, M. T. (2017). Standardizing size-and shape-controlled synthesis of monodisperse magnetite (Fe3O4) nanocrystals by identifying and exploiting effects of organic impurities. ACS nano, 11(6), 6370–6381. https://doi.org/10.1021/acsnano.7b02752 DOI: https://doi.org/10.1021/acsnano.7b02752
Mathew, D. S., & Juang, R. S. (2007). An overview of the structure and magnetism of spinel ferrite nanoparticles and their synthesis in microemulsions. Chemical engineering journal, 129(1-3), 51–65. https://doi.org/10.1016/j.cej.2006.11.001 DOI: https://doi.org/10.1016/j.cej.2006.11.001
Lu, A. H., Salabas, E. E., & Schüth, F. (2007). Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angewandte Chemie International Edition, 46(8), 1222–1244. https://doi.org/10.1002/anie.200602866 DOI: https://doi.org/10.1002/anie.200602866
Peng, S., & Sun, S. (2007). Synthesis and characterization of monodisperse hollow Fe3O4 nanoparticles. Angewandte Chemie International Edition, 46(22), 4155–4158. https://doi.org/10.1002/anie.200700677 DOI: https://doi.org/10.1002/anie.200700677
Schwaminger, S. P., Syhr, C., & Berensmeier, S. (2020). Controlled synthesis of magnetic iron oxide nanoparticles: magnetite or maghemite? Crystals, 10(3), 214. https://doi.org/10.3390/cryst10030214 DOI: https://doi.org/10.3390/cryst10030214
Reindl, M., Zach, V., Cvirn, G., & Schwaminger, S. P. (2025). Influence of coprecipitation synthesis parameters on the physicochemical properties and biological effects of iron oxide nanoparticles. Nanoscale Advances, 7(22), 7395–7407. https://doi.org/10.1039/d5na00632e DOI: https://doi.org/10.1039/D5NA00632E
Schwaminger, S. P., Bauer, D., Fraga-García, P., Wagner, F. E., & Berensmeier, S. (2017). Oxidation of magnetite nanoparticles NanoNPs: impact on surface and crystal properties. CrystEngComm, 19(2), 246–255. https://doi.org/10.1039/C6CE02421A DOI: https://doi.org/10.1039/C6CE02421A
Siregar, N., Indrayana, I. P. T., Suharyadi, E., Kato, T., & Iwata, S. (2017). Effect of synthesis temperature and NaOH concentration on microstructural and magnetic properties of Mn0.5Zn0.5Fe2O4 nanoparticles. In IOP Conference Series: Materials Science and Engineering, 202(1), 012048. https://doi.org/10.1088/1757-899X/202/1/012048 DOI: https://doi.org/10.1088/1757-899X/202/1/012048
Ghazanfari, M. R., Kashefi, M., & Jaafari, M. R. (2016). Optimizing and modeling of effective parameters on the structural and magnetic properties of Fe3O4 nanoparticles synthesized by coprecipitation technique using response surface methodology. Journal of Magnetism and Magnetic Materials, 409, 134–142. https://doi.org/10.1016/j.jmmm.2016.02.094 DOI: https://doi.org/10.1016/j.jmmm.2016.02.094
Valenzuela, R., Fuentes, M. C., Parra, C., Baeza, J., Duran, N., Sharma, S. K., ... & Freer, J. (2009). Influence of stirring velocity on the synthesis of magnetite nanoparticles (Fe3O4) by the co-precipitation method. Journal of Alloys and Compounds, 488(1), 227–231. https://doi.org/10.1016/j.jallcom.2009.08.087 DOI: https://doi.org/10.1016/j.jallcom.2009.08.087
Huang, Z., Tang, F., & Zhang, L. (2005). Morphology control and texture of Fe3O4 nanoparticle-coated polystyrene microspheres by ethylene glycol in forced hydrolysis reaction. Thin Solid Films, 471(1-2), 105–112. https://doi.org/10.1016/j.tsf.2004.04.042 DOI: https://doi.org/10.1016/j.tsf.2004.04.042
Furusawa, K., Norde, W. & Lyklema, J. (1972). A method for preparing surfactant-free polystyrene latices of high surface charge. Kolloid-Zeitschrift & Zeitschrift für Polymere, 250(9), 908–909. https://doi.org/10.1007/BF01506246 DOI: https://doi.org/10.1007/BF01506246
Sobenin, D.V., Solovov, R.D. & Ershov B.G. (2026). Transformation of zerovalent iron nanoparticles in isopropanol into oxide particles: effect of water and air on mechanism and kinetics. Journal of Molecular Liquids, 447, 129333. https://doi.org/10.1016/j.molliq.2026.129333 DOI: https://doi.org/10.1016/j.molliq.2026.129333
Elmore, W.C. (1938). Ferromagnetic colloid for studying magnetic structures. Physical Review, 54(4), 309–310. https://doi.org/10.1103/PhysRev.54.309 DOI: https://doi.org/10.1103/PhysRev.54.309
Xu, N., Yan, H., Jiao, X., Jiang, L., Zhang, R., Wang, J., Liu, Z., Liu, Z., Gu, Y., Gang, F., Wang, X., Zhao, L., & Sun, X. (2020). Effect of OH− concentration on Fe3O4 nanoparticle morphologies supported by first principle calculation. Journal of Crystal Growth, 547, 125780. https://doi.org/10.1016/j.jcrysgro.2020.125780 DOI: https://doi.org/10.1016/j.jcrysgro.2020.125780
Bhagwat, S., Singh, H., Athawale, A., Hannoyer, B., Jouen, S., Lefez, B., Kundaliya, D., Pasricha, R., Kulkarni, S., & Ogale, S. (2007). Low temperature synthesis of magnetite and maghemite nanoparticles. Journal of Nanoscience and Nanotechnology, 7(12), 4294–4302. https://doi.org/10.1166/jnn.2007.873 DOI: https://doi.org/10.1166/jnn.2007.873
Vinila, V.S.; Isac, J. (2022). Chapter 14 — Synthesis and structural studies of superconducting perovskite GdBa2Ca3Cu4O10.5+δ nanosystems. Design, Fabrication, and Characterization of Multifunctional Nanomaterials, 319–341. https://doi.org/10.1016/B978-0-12-820558-7.00022-4 DOI: https://doi.org/10.1016/B978-0-12-820558-7.00022-4
Frison, R., Cernuto, G., Cervellino, A., Zaharko, O., Colonna, G. M., Guagliardi, A., Masciocchi, N. (2013). Magnetite–Maghemite Nanoparticles in the 5–15 Nm Range: Correlating the Core–Shell Composition and the Surface Structure to the Magnetic Properties. A Total Scattering Study. Chemistry of Materials, 25(23), 4820–4827. https://doi.org/10.1021/cm403360f. DOI: https://doi.org/10.1021/cm403360f
Dzeranov, A., Bondarenko, L., Saman, D., Prokof’ev, M., Terekhova, V., Telegina, L., Dzhardimalieva, G., Bolotskaya, S., Kydralieva, K. (2024). Effects of water induced aging on iron (oxyhydr)oxides nanoparticles: linking crystal structure, iron ion release, and toxicity. Chemical Papers. https://doi.org/10.1007/s11696-024-03373-x. DOI: https://doi.org/10.21203/rs.3.rs-3571668/v1
Bondarenko, L., Kahru, A., Terekhova, V., Dzhardimalieva, G., Uchanov, P., Kydralieva, K. (2020). Effects of Humic Acids on the Ecotoxicity of Fe3O4 Nanoparticles and Fe-Ions: Impact of Oxidation and Aging. Nanomaterials, 10(10), 2011. https://doi.org/10.3390/nano10102011 DOI: https://doi.org/10.3390/nano10102011
Chen, L., Ma, J., Li, X., Zhang, J., Fang, J., Guan, Y., & Xie, P. (2011). Strong enhancement on fenton oxidation by addition of hydroxylamine to accelerate the ferric and ferrous iron cycles. Environmental science & technology, 45(9), 3925–3930. https://doi.org/10.1021/es2002748 DOI: https://doi.org/10.1021/es2002748
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Artur A. Dzeranov, Lyubov S. Bondarenko, Michail V. Prokofiev, Roman А. Bondarenko, Danil R. Abramov, Gulzhian I. Dzhardimalieva, Kamila A. Kydralieva

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



