Comparative Development and Characterization of Itraconazole-Loaded Solid Lipid Nanoparticles Incorporating Myristic Acid and Pluronic F127 for Oral Delivery
DOI:
https://doi.org/10.31489/2959-0663/1-26-11Keywords:
itraconazole, solid lipid nanoparticles, solubility enhancement, stearic acid, myristic acid, Pluronic F127, Tween 80, controlled drug releaseAbstract
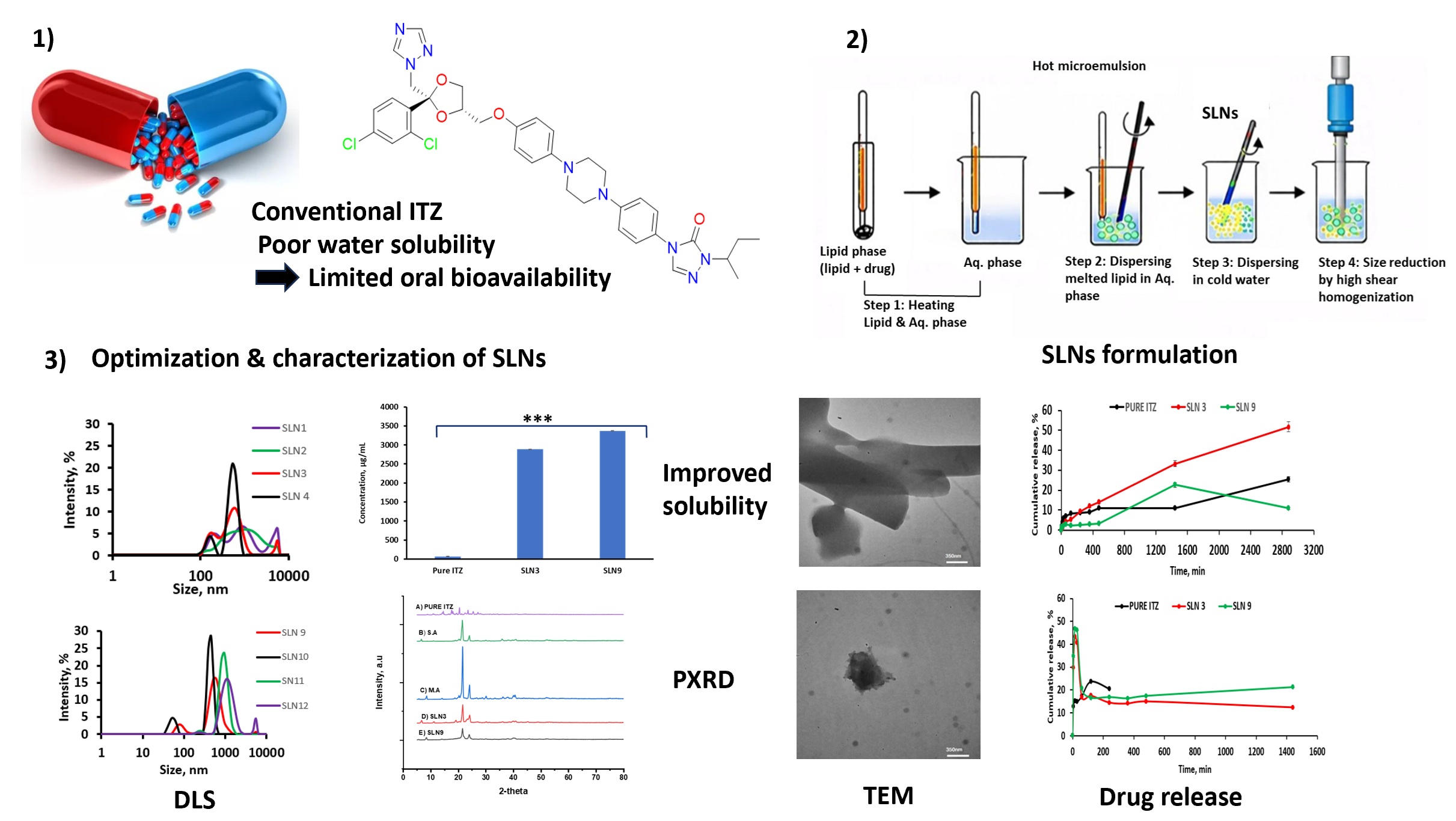
This study developed itraconazole-loaded solid lipid nanoparticles (SLNs) to enhance the solubility of this poorly water-soluble antifungal drug and evaluate key physicochemical properties. SLNs were prepared using the microemulsion technique with solid lipids stearic acid, palmitic acid, and myristic acid, and surfactants Tween 80 and Pluronic F127. The synthesized SLNs were characterized using dynamic light scattering (DLS) and electrophoretic light scattering (ELS) for size and zeta potential determination, while transmission electron microscopy (TEM) and field emission scanning electron microscopy (FESEM) were employed to examine surface morphology. Furthermore, the structural and thermal properties of the formulation were analyzed via Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), and differential scanning calorimetry (DSC). Among the formulations, SLN3 (containing stearic acid–Pluronic F127) and SLN9 (containing myristic acid–Tween 80) exhibited the smallest particle sizes and lowest polydispersity indices. Encapsulation efficiency was 97.04 ± 0.004 % for SLN3 and 42.69 ± 0.02 % for SLN9, with drug loading capacities of 3 ± 0.1 % and 1.8 ± 0.17 %, and yields of 50.03 ± 3.55 % and 57.9 ± 6.6 %, respectively. Solubility of ITZ increased to 2900 µg/mL (SLN3) and 3369 µg/mL (SLN9). In vitro release studies demonstrated controlled and sustained drug release, with SLNs exhibiting formulation- and pH-dependent behavior; SLN3 provided more prolonged release under acidic conditions, whereas SLN9 showed relatively higher release at intestinal pH, reflecting differences in lipid chain length and surfactant type. These results indicate that the optimized SLNs improve ITZ solubility and exhibit favorable physicochemical characteristics, supporting their potential as oral delivery systems for poorly soluble antifungal agents.
References
Alam, S., Iqbal, Z., Ali, A., Khar, R. K., Ahmad, F. J., Akhter, S., & Talegaonkar, S. (2009). Microemulsion as a Potential Transdermal Carrier for Poorly Water Soluble Antifungal Drug Itraconazole. Journal of Dispersion Science and Technology, 31(1), 84–94. https://doi.org/10.1080/01932690903107265 DOI: https://doi.org/10.1080/01932690903107265
Chhatbar, M., Borkhataria, C., Patel, O., Raichura, K., Pethani, T., Parmar, G., Mori, D., & Manek, R. (2025). Enhancing the solubility and bioavailability of itraconazole through pharmaceutical cocrystallization: A promising strategy for drug formulation. Journal of Pharmaceutical Sciences, 114(6), 103770. https://doi.org/10.1016/j.xphs.2025.103770 DOI: https://doi.org/10.1016/j.xphs.2025.103770
Pardeike, J., Weber, S., Haber, T., Wagner, J., Zarfl, H. P., Plank, H., & Zimmer, A. (2011). Development of an itraconazole-loaded nanostructured lipid carrier (NLC) formulation for pulmonary application. International Journal of Pharmaceutics, 419(1–2), 329–338. https://doi.org/10.1016/j.ijpharm.2011.07.040 DOI: https://doi.org/10.1016/j.ijpharm.2011.07.040
Osborn, M. R., Zuniga-Moya, J. C., Mazi, P. B., Rauseo, A. M., & Spec, A. (2025). Side effects associated with itraconazole therapy. The Journal of Antimicrobial Chemotherapy, 80(2), 503–508. https://doi.org/10.1093/jac/dkae437 DOI: https://doi.org/10.1093/jac/dkae437
Cheng, Y., Xu, Z., Ma, M., & Xu, T. (2008). Dendrimers as drug carriers: Applications in different routes of drug administration. Journal of Pharmaceutical Sciences, 97(1), 123–143. https://doi.org/10.1002/jps.21079 DOI: https://doi.org/10.1002/jps.21079
Hu, L., Tang, X., & Cui, F. (2004). Solid lipid nanoparticles (SLNs) to improve oral bioavailability of poorly soluble drugs. The Journal of Pharmacy and Pharmacology, 56(12), 1527–1535. https://doi.org/10.1211/0022357044959 DOI: https://doi.org/10.1211/0022357044959
Xie, S., Zhu, L., Dong, Z., Wang, Y., Wang, X., & Zhou, W. (2011). Preparation and evaluation of ofloxacin-loaded palmitic acid solid lipid nanoparticles. International Journal of Nanomedicine, 6, 547–555. https://doi.org/10.2147/IJN.S17083 DOI: https://doi.org/10.2147/IJN.S17083
Severino, P., Andreani, T., Macedo, A. S., Fangueiro, J. F., Santana, M. H. A., Silva, A. M., & Souto, E. B. (2012). Current State-of-Art and New Trends on Lipid Nanoparticles (SLN and NLC) for Oral Drug Delivery. Journal of Drug Delivery, 2012, 750891. https://doi.org/10.1155/2012/750891 DOI: https://doi.org/10.1155/2012/750891
Dattani, S., Li, X., Lampa, C., Lechuga-Ballesteros, D., Barriscale, A., Damadzadeh, B., & Jasti, B. R. (2023). A comparative study on micelles, liposomes and solid lipid nanoparticles for paclitaxel delivery. International Journal of Pharmaceutics, 631, 122464. https://doi.org/10.1016/j.ijpharm.2022.122464 DOI: https://doi.org/10.1016/j.ijpharm.2022.122464
Wissing, S. A., & Müller, R. H. (2003). The influence of solid lipid nanoparticles on skin hydration and viscoelasticity — In vivo study. European Journal of Pharmaceutics and Biopharmaceutics: Official Journal of Arbeitsgemeinschaft Fur Pharmazeutische Verfahrenstechnik e.V, 56(1), 67–72. https://doi.org/10.1016/s0939-6411(03)00040-7 DOI: https://doi.org/10.1016/S0939-6411(03)00040-7
Kumar, N., & Goindi, S. (2021). Development and Optimization of Itraconazole-Loaded Solid Lipid Nanoparticles for Topical Administration Using High Shear Homogenization Process by Design of Experiments: In Vitro, Ex Vivo and In Vivo Evaluation. AAPS PharmSciTech, 22(7), 248. https://doi.org/10.1208/s12249-021-02118-3 DOI: https://doi.org/10.1208/s12249-021-02118-3
Mukherjee, S., Ray, S., & Thakur, R. S. (2009). Design and evaluation of itraconazole loaded solid lipid nanoparticulate system for improving the antifungal therapy. Pakistan Journal of Pharmaceutical Sciences, 22(2), 131–138.
Müller, R. H., Shegokar, R., & Keck, C. M. (2011). 20 years of lipid nanoparticles (SLN and NLC): Present state of development and industrial applications. Current Drug Discovery Technologies, 8(3), 207–227. https://doi.org/10.2174/157016311796799062 DOI: https://doi.org/10.2174/157016311796799062
Mehnert, W., & Mäder, K. (2012). Solid lipid nanoparticles: Production, characterization and applications. Advanced Drug Delivery Reviews, 64, 83–101. https://doi.org/10.1016/j.addr.2012.09.021 DOI: https://doi.org/10.1016/j.addr.2012.09.021
Mishra, V., Bansal, K. K., Verma, A., Yadav, N., Thakur, S., Sudhakar, K., & Rosenholm, J. M. (2018). Solid Lipid Nanoparticles: Emerging Colloidal Nano Drug Delivery Systems. Pharmaceutics, 10(4), 191. https://doi.org/10.3390/pharmaceutics10040191 DOI: https://doi.org/10.3390/pharmaceutics10040191
Cassano, R., Ferrarelli, T., Mauro, M. V., Cavalcanti, P., Picci, N., & Trombino, S. (2016). Preparation, characterization and in vitro activities evaluation of solid lipid nanoparticles based on PEG-40 stearate for antifungal drugs vaginal delivery. Drug Delivery, 23(3), 1047–1056. https://doi.org/10.3109/10717544.2014.932862 DOI: https://doi.org/10.3109/10717544.2014.932862
Shah, V., Lesko, L., Fan, J., Fleischer, N., Handerson, J., Malinowski, H., Makary, M., Ouderkirk, L., Roy, S., Sathe, P., Singh, G., Tillman, L., Tsong, Y., & Williams, R. (1997). fDA Guidance for Industry 1 Dissolution Testing of Immediate Release Solid Oral Dosage Forms. Dissolution Technologies, 4, 15–22. https://doi.org/10.14227/DT040497P15 DOI: https://doi.org/10.14227/DT040497P15
Zoubari, G., Staufenbiel, S., Volz, P., Alexiev, U., & Bodmeier, R. (2017). Effect of drug solubility and lipid carrier on drug release from lipid nanoparticles for dermal delivery. European Journal of Pharmaceutics and Biopharmaceutics: Official Journal of Arbeitsgemeinschaft Fur Pharmazeutische Verfahrenstechnik e.V, 110, 39–46. https://doi.org/10.1016/j.ejpb.2016.10.021 DOI: https://doi.org/10.1016/j.ejpb.2016.10.021
Gaba, B., Fazil, M., Khan, S., Ali, A., Baboota, S., & Ali, J. (2015). Nanostructured lipid carrier system for topical delivery of terbinafine hydrochloride. Bulletin of Faculty of Pharmacy, Cairo University, 53(2), 147–159. https://doi.org/10.1016/j.bfopcu.2015.10.001 DOI: https://doi.org/10.1016/j.bfopcu.2015.10.001
Khalil, R. M., Abd El-Bary, A., Kassem, M. A., Ghorab, M. M., & Basha, M. (2013). Influence of formulation parameters on the physicochemical properties of meloxicam-loaded solid lipid nanoparticles. Egyptian Pharmaceutical Journal, 12(1), 63. https://doi.org/10.7123/01.EPJ.0000428643.74323.d9
Elmowafy, M., & Al-Sanea, M. M. (2021). Nanostructured lipid carriers (NLCs) as drug delivery platform: Advances in formulation and delivery strategies. Saudi Pharmaceutical Journal, 29(9), 999–1012. https://doi.org/10.1016/j.jsps.2021.07.015 DOI: https://doi.org/10.1016/j.jsps.2021.07.015
Zimmermann, E., & Müller, R. H. (2001). Electrolyte- and pH-stabilities of aqueous solid lipid nanoparticle (SLNTM) dispersions in artificial gastrointestinal media. European Journal of Pharmaceutics and Biopharmaceutics, 52(2), 203–210. https://doi.org/10.1016/S0939-6411(01)00167-9 DOI: https://doi.org/10.1016/S0939-6411(01)00167-9
Sanna, V., Gavini, E., Cossu, M., Rassu, G., & Giunchedi, P. (2007). Solid lipid nanoparticles (SLN) as carriers for the topical delivery of econazole nitrate: In-vitro characterization, ex-vivo and in-vivo studies. The Journal of Pharmacy and Pharmacology, 59(8), 1057–1064. https://doi.org/10.1211/jpp.59.8.0002 DOI: https://doi.org/10.1211/jpp.59.8.0002
Younus, M., Hawley, A., Boyd, B. J., & Rizwan, S. B. (2018). Bulk and dispersed aqueous behaviour of an endogenous lipid, selachyl alcohol: Effect of Tween 80 and Pluronic F127 on nanostructure. Colloids and Surfaces. B, Biointerfaces, 169, 135–142. https://doi.org/10.1016/j.colsurfb.2018.05.013 DOI: https://doi.org/10.1016/j.colsurfb.2018.05.013
Karjiban, R. A., Basri, M., Rahman, M. B. A., & Salleh, A. B. (2012). Structural Properties of Nonionic Tween80 Micelle in Water Elucidated by Molecular Dynamics Simulation. APCBEE Procedia, 2nd International Conference on Chemistry and Chemical Process (ICCCP 2012) May 5-6, 2012, 3, 287–297. https://doi.org/10.1016/j.apcbee.2012.06.084 DOI: https://doi.org/10.1016/j.apcbee.2012.06.084
Kumar, M., Tiwari, A., Asdaq, S. M. B., Nair, A. B., Bhatt, S., Shinu, P., Al Mouslem, A. K., Jacob, S., Alamri, A. S., Alsanie, W. F., Alhomrani, M., Tiwari, V., Devi, S., Pathania, A., & Sreeharsha, N. (2022). Itraconazole loaded nano-structured lipid carrier for topical ocular delivery: Optimization and evaluation. Saudi Journal of Biological Sciences, 29(1), 1–10. https://doi.org/10.1016/j.sjbs.2021.11.006 DOI: https://doi.org/10.1016/j.sjbs.2021.11.006
Ebrahimi, H. A., Javadzadeh, Y., Hamidi, M., & Jalali, M. B. (2015). Repaglinide-loaded solid lipid nanoparticles: Effect of using different surfactants/stabilizers on physicochemical properties of nanoparticles. Daru: Journal of Faculty of Pharmacy, Tehran University of Medical Sciences, 23(1), 46. https://doi.org/10.1186/s40199-015-0128-3 DOI: https://doi.org/10.1186/s40199-015-0128-3
Garud, A., Singh, D., & Garud, N. (2012). Solid Lipid Nanoparticles (SLN): Method, Characterization and Applications. International Current Pharmaceutical Journal, 1(11), 384–393. https://doi.org/10.3329/icpj.v1i11.12065 DOI: https://doi.org/10.3329/icpj.v1i11.12065
Pizzol, C. D., Filippin-Monteiro, F. B., Restrepo, J. A. S., Pittella, F., Silva, A. H., Alves de Souza, P., Machado de Campos, A., & Creczynski-Pasa, T. B. (2014). Influence of Surfactant and Lipid Type on the Physicochemical Properties and Biocompatibility of Solid Lipid Nanoparticles. International Journal of Environmental Research and Public Health, 11(8), 8581–8596. https://doi.org/10.3390/ijerph110808581 DOI: https://doi.org/10.3390/ijerph110808581
Mellaerts, R., Mols, R., Jammaer, J. A. G., Aerts, C. A., Annaert, P., Van Humbeeck, J., Van den Mooter, G., Augustijns, P., & Martens, J. A. (2008). Increasing the oral bioavailability of the poorly water soluble drug itraconazole with ordered mesoporous silica. European Journal of Pharmaceutics and Biopharmaceutics: Official Journal of Arbeitsgemeinschaft Fur Pharmazeutische Verfahrenstechnik e.V, 69(1), 223–230. https://doi.org/10.1016/j.ejpb.2007.11.006 DOI: https://doi.org/10.1016/j.ejpb.2007.11.006
Mohanty, B., Majumdar, D. K., Mishra, S. K., Panda, A. K., & Patnaik, S. (2015). Development and characterization of itraconazole-loaded solid lipid nanoparticles for ocular delivery. Pharmaceutical Development and Technology, 20(4), 458–464. https://doi.org/10.3109/10837450.2014.882935 DOI: https://doi.org/10.3109/10837450.2014.882935
Pandey, S., Shaikh, F., Gupta, A., Tripathi, P., & Yadav, J. S. (2022). A Recent Update: Solid Lipid Nanoparticles for Effective Drug Delivery. Advanced Pharmaceutical Bulletin, 12(1), 17–33. https://doi.org/10.34172/apb.2022.007 DOI: https://doi.org/10.34172/apb.2022.007
Nesseem, D. I. (2001). Formulation and evaluation of itraconazole via liquid crystal for topical delivery system. Journal of Pharmaceutical and Biomedical Analysis, 26(3), 387–399. https://doi.org/10.1016/s0731-7085(01)00414-9 DOI: https://doi.org/10.1016/S0731-7085(01)00414-9
Subroto, E., Afifah, T. N., Harlina, P. W., Indiarto, R., Pangawikan, A. D., Huda, S., Wiguna, B., & Geng, F. (2025). Solid lipid nanoparticles of mangosteen peel extract based on monoacylglycerol-diacylglycerol-rich fat and stearic acid: Study on physicochemical properties and encapsulation efficiency. Future Foods, 12, 100719. https://doi.org/10.1016/j.fufo.2025.100719 DOI: https://doi.org/10.1016/j.fufo.2025.100719
Tao, T., Zhao, Y., Wu, J., & Zhou, B. (2009). Preparation and evaluation of itraconazole dihydrochloride for the solubility and dissolution rate enhancement. International Journal of Pharmaceutics, 367(1–2), 109–114. https://doi.org/10.1016/j.ijpharm.2008.09.034 DOI: https://doi.org/10.1016/j.ijpharm.2008.09.034
Triboandas, H., Pitt, K., Bezerra, M., Ach-Hubert, D., & Schlindwein, W. (2022). Itraconazole Amorphous Solid Dispersion Tablets: Formulation and Compaction Process Optimization Using Quality by Design Principles and Tools. Pharmaceutics, 14(11), 2398. https://doi.org/10.3390/pharmaceutics14112398 DOI: https://doi.org/10.3390/pharmaceutics14112398
Franken, L. E., Boekema, E. J., & Stuart, M. C. A. (2017). Transmission Electron Microscopy as a Tool for the Characterization of Soft Materials: Application and Interpretation. Advanced Science, 4(5), 1600476. https://doi.org/10.1002/advs.201600476 DOI: https://doi.org/10.1002/advs.201600476
Nogueira, S. S., Samaridou, E., Simon, J., Frank, S., Beck-Broichsitter, M., & Mehta, A. (2024). Analytical techniques for the characterization of nanoparticles for mRNA delivery. European Journal of Pharmaceutics and Biopharmaceutics, 198, 114235. https://doi.org/10.1016/j.ejpb.2024.114235 DOI: https://doi.org/10.1016/j.ejpb.2024.114235
Filippov, S. K., Khusnutdinov, R., Murmiliuk, A., Inam, W., Zakharova, L. Ya., Zhang, H., & Khutoryanskiy, V. V. (2023). Dynamic light scattering and transmission electron microscopy in drug delivery: a roadmap for correct characterization of nanoparticles and interpretation of results. Materials Horizons, 10(12), 5354–5370. https://doi.org/10.1039/d3mh00717k DOI: https://doi.org/10.1039/D3MH00717K
Subroto, E., Andoyo, R., Indiarto, R., Wulandari, E., & Wadhiah, E. F. N. (2022). Preparation of Solid Lipid Nanoparticle-Ferrous Sulfate by Double Emulsion Method Based on Fat Rich in Monolaurin and Stearic Acid. Nanomaterials, 12(17), 3054. https://doi.org/10.3390/nano12173054 DOI: https://doi.org/10.3390/nano12173054
Naseri, N., Valizadeh, H., & Zakeri-Milani, P. (2015). Solid Lipid Nanoparticles and Nanostructured Lipid Carriers: Structure, Preparation and Application. Advanced Pharmaceutical Bulletin, 5(3), 305–313. https://doi.org/10.15171/apb.2015.043 DOI: https://doi.org/10.15171/apb.2015.043
Chen, W., Gu, B., Wang, H., Pan, J., Lu, W., & Hou, H. (2008). Development and evaluation of novel itraconazole-loaded intravenous nanoparticles. International Journal of Pharmaceutics, 362(1–2), 133–140. https://doi.org/10.1016/j.ijpharm.2008.05.039 DOI: https://doi.org/10.1016/j.ijpharm.2008.05.039
Das, S., Ng, W. K., Kanaujia, P., Kim, S., & Tan, R. B. H. (2011). Formulation design, preparation and physicochemical characterizations of solid lipid nanoparticles containing a hydrophobic drug: Effects of process variables. Colloids and Surfaces. B, Biointerfaces, 88(1), 483–489. https://doi.org/10.1016/j.colsurfb.2011.07.036 DOI: https://doi.org/10.1016/j.colsurfb.2011.07.036
Lang, B., McGinity, J. W., & Williams, R. O. (2014). Dissolution enhancement of itraconazole by hot-melt extrusion alone and the combination of hot-melt extrusion and rapid freezing — Effect of formulation and processing variables. Molecular Pharmaceutics, 11(1), 186–196. https://doi.org/10.1021/mp4003706 DOI: https://doi.org/10.1021/mp4003706
Salah, E., Abouelfetouh, M. M., Pan, Y., Chen, D., & Xie, S. (2020). Solid lipid nanoparticles for enhanced oral absorption: A review. Colloids and Surfaces. B, Biointerfaces, 196, 111305. https://doi.org/10.1016/j.colsurfb.2020.111305 DOI: https://doi.org/10.1016/j.colsurfb.2020.111305
Silva, A. C., González-Mira, E., García, M. L., Egea, M. A., Fonseca, J., Silva, R., Santos, D., Souto, E. B., & Ferreira, D. (2011). Preparation, characterization and biocompatibility studies on risperidone-loaded solid lipid nanoparticles (SLN): High pressure homogenization versus ultrasound. Colloids and Surfaces. B, Biointerfaces, 86(1), 158–165. https://doi.org/10.1016/j.colsurfb.2011.03.035 DOI: https://doi.org/10.1016/j.colsurfb.2011.03.035
Ghasemiyeh, P., & Mohammadi-Samani, S. (2018). Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Research in Pharmaceutical Sciences, 13(4), 288–303. https://doi.org/10.4103/1735-5362.235156 DOI: https://doi.org/10.4103/1735-5362.235156
Zainol, S., Basri, M., Basri, H. B., Shamsuddin, A. F., Abdul-Gani, S. S., Karjiban, R. A., & Abdul-Malek, E. (2012). Formulation Optimization of a Palm-Based Nanoemulsion System Containing Levodopa. International Journal of Molecular Sciences, 13(10), 13049–13064. https://doi.org/10.3390/ijms131013049 DOI: https://doi.org/10.3390/ijms131013049
Andonova, V., & Peneva, P. (2017). Characterization Methods for Solid Lipid Nanoparticles (SLN) and Nanostructured Lipid Carriers (NLC). Current Pharmaceutical Design. https://doi.org/10.2174/1381612823666171115105721 DOI: https://doi.org/10.2174/1381612823666171115105721
Martins, S., Tho, I., Ferreira, D. C., Souto, E. B., & Brandl, M. (2011). Physicochemical properties of lipid nanoparticles: Effect of lipid and surfactant composition. Drug Development and Industrial Pharmacy, 37(7), 815–824. https://doi.org/10.3109/03639045.2010.545414 DOI: https://doi.org/10.3109/03639045.2010.545414
Stahl, M. A., Lüdtke, F. L., Grimaldi, R., Gigante, M. L., & Ribeiro, A. P. B. (2024). Characterization and stability of solid lipid nanoparticles produced from different fully hydrogenated oils. Food Research International (Ottawa, Ont.), 176, 113821. https://doi.org/10.1016/j.foodres.2023.113821 DOI: https://doi.org/10.1016/j.foodres.2023.113821
Kawakami, K., Oda, N., Miyoshi, K., Funaki, T., & Ida, Y. (2006). Solubilization behavior of a poorly soluble drug under combined use of surfactants and cosolvents. European Journal of Pharmaceutical Sciences: Official Journal of the European Federation for Pharmaceutical Sciences, 28(1–2), 7–14. https://doi.org/10.1016/j.ejps.2005.11.012 DOI: https://doi.org/10.1016/j.ejps.2005.11.012
Segale, L., Giovannelli, L., Mannina, P., & Pattarino, F. (2015). Formulation and characterization study of itraconazole-loaded microparticles. Pharmaceutical Development and Technology, 20(2), 153–158. https://doi.org/10.3109/10837450.2013.852572 DOI: https://doi.org/10.3109/10837450.2013.852572
Lee, J.-H., Park, C., Weon, K.-Y., Kang, C.-Y., Lee, B.-J., & Park, J.-B. (2021). Improved Bioavailability of Poorly Water-Soluble Drug by Targeting Increased Absorption through Solubility Enhancement and Precipitation Inhibition. Pharmaceuticals (Basel, Switzerland), 14(12), 1255. https://doi.org/10.3390/ph14121255 DOI: https://doi.org/10.3390/ph14121255
Parikh, T., Sandhu, H. K., Talele, T. T., & Serajuddin, A. T. M. (2016). Characterization of Solid Dispersion of Itraconazole Prepared by Solubilization in Concentrated Aqueous Solutions of Weak Organic Acids and Drying. Pharmaceutical Research, 33(6), 1456–1471. https://doi.org/10.1007/s11095-016-1890-8 DOI: https://doi.org/10.1007/s11095-016-1890-8
Kim, J.-K., Park, J.-S., & Kim, C.-K. (2010). Development of a binary lipid nanoparticles formulation of itraconazole for parenteral administration and controlled release. International Journal of Pharmaceutics, 383(1–2), 209–215. https://doi.org/10.1016/j.ijpharm.2009.09.008 DOI: https://doi.org/10.1016/j.ijpharm.2009.09.008
Khan, J., Rades, T., & Boyd, B. J. (2016). Lipid-Based Formulations Can Enable the Model Poorly Water-Soluble Weakly Basic Drug Cinnarizine To Precipitate in an Amorphous-Salt Form During In Vitro Digestion. Molecular Pharmaceutics, 13(11), 3783–3793. https://doi.org/10.1021/acs.molpharmaceut.6b00594 DOI: https://doi.org/10.1021/acs.molpharmaceut.6b00594
Das, S., & Chaudhury, A. (2011). Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech, 12(1), 62–76. https://doi.org/10.1208/s12249-010-9563-0 DOI: https://doi.org/10.1208/s12249-010-9563-0
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Darya Wahhab Kareem, Twana Mohammed M.Ways

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



