Benzodioxole Scaffold Incorporating Compound with Promising Anti-Fungal Potential: An Overview
DOI:
https://doi.org/10.31489/2959-0663/1-25-1Keywords:
Heterocyclic, 1,3-benzodioxole, fungal infection, biophoric nature, phytopathogenic, human pathogenic, Pharmacological properties, antifungal therapyAbstract
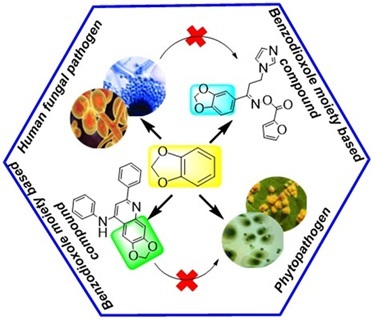
This review focuses on the compounds with 1,3-benzodioxole scaffold and their antifungal potential. Different online sources including Google Scholar, Pubchem and Science Direct were used to collect information on benzodioxole containing compounds as antifungal agents from articles published between 2003 and 2022. The health threat posed by fungal infections to humans makes an ongoing search for more different antifungal agents necessary. Diverse heterocyclic moiety proves beneficial in fungal infection, among all of them benzodioxole incorporation into the compound was also found to be effective. The 1,3-benzodioxole or methylenedioxy benzene scaffold is highly adaptable, enabling a range of chemical modifications. The biophoric nature of this scaffold imparts distinctive pharmacological properties, contributing to its broad-spectrum activity. It was shown that the addition of heterocyclic moiety (e.g. primidinone, imidazole, thiazole, etc.), aliphatic linker, amide linker at position 5 and furthermore electron withdrawing group at position 6 enhances the antifungal potential against various phytopathogenic and human pathogenic fungi species. This review highlights the anti-fungal status of benzodioxole scaffold-containing compounds focusing on their efficacy against both phytopathogenic and human pathogenic fungal species. Additionally, the review discusses the structural modification and the future prospects of these compounds in antifungal therapy.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Amita Joshi Rana , Sanjana Bisht , Kumud Upadhyaya, Pawan Singh, Mahendra Rana, Shweta Singh

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



