Multicomponent Synthesis of Novel Unsymmetric 6-Aryl Substituted 5-Nitropyridines
DOI:
https://doi.org/10.31489/2959-0663/2-24-3Keywords:
green chemistry, multicomponent reaction, substituted 2-nitroacetophenones, pyridine derivatives, 5(3)-nitropyridines, 5(3)-nitro-1,4-dihydropyridines, 3-acetyl-5-nitropyridines , heterocyclic compoundsAbstract
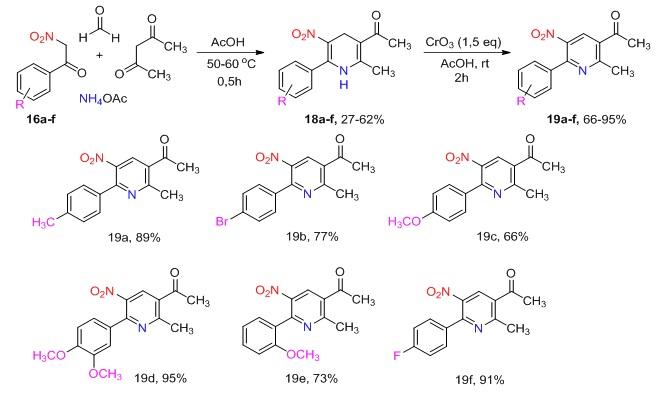
We have previously studied a multicomponent reaction for the synthesis of unsymmetrical 5-nitro-1,4-dihydropyridines using unsubstituted 2-nitroacetophenone, 1,3-dicarbonyl compounds, and various aldehydes such as formaldehyde, acetaldehyde, and furfural. This paper reports the use of unsymmetrical 3-acetyl-5-nitro-1,4-dihydropyridines containing aryl substituents at the 6-position in a multicomponent synthesis reaction. The starting aryl-substituted nitroacetophenones were prepared by two methods. The first method involved the two-step Katritzky method, which is described in the literature. This method consists of preparing N-acylbenzotriazoles from the corresponding substituted derivatives of benzoic acid and 1,2,3-benzotriazole in the presence of thionyl chloride. This is followed by C-acylation of nitromethane in supernatron medium (t-BuOK – DMSO). A number of 2-nitroacetophenone derivatives were prepared from more commercially available aromatic aldehydes by the Henry reaction with nitromethane followed by oxidation of the resulting secondary nitroalcohols. The multicomponent reaction of 6-aryl-substituted 5-nitro-1,4-dihydropyridines and their subsequent aromatization into 5-nitropyridines allowed us to reduce the overall reaction time by more than 40 times and to increase the total yield of 5-nitro-6-arylpyridines by an average of twofold compared to the method described in the literature. Furthermore, the 3-acetyl-5-nitropyridines we have obtained are significant intermediates in the synthesis of novel, more complex heterocyclic systems with potential biological activity. These systems include δ-carbolines and epoxybenzooxocyno[4,3-b]pyridines, which are currently of great interest for the study of their properties.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Ilya Kulakov, Semyon Chikunov, Andrey Elyshev, Ivan Kulakov

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



