Effect of Acetylene Properties on its Gas Sensing by NiO Doped ZnO Clusters: A Transition State Theory Model
DOI:
https://doi.org/10.31489/2959-0663/4-24-8Keywords:
NiO-doping, ZnO cluster, Acetylene gas sensor, Density functional theory, Transition state, autoignition , thermodynamic quantities, B3LYP, GD3BJAbstract
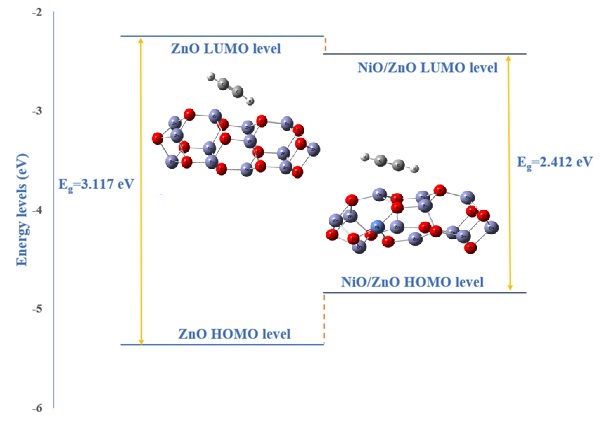
The sensitivity of nickel oxide doped zinc oxide to industrial gas acetylene was calculated and compared to available experimental data. The adsorption of C2H2 on the NiO-doped ZnO surface and the activation transition states formed afterward were studied computationally. B3LYP version of density functional theory with 6-311G** basis set, including dispersion correction (GD3BJ) was performed for the calculations with the help of Gaussian 09 software. Thermodynamic quantities of the reaction of C2H2 with NiO-doped ZnO surfaces, such as Gibbs free energy, enthalpy, and entropy, were used to interpret the reaction at the temperature range 25–325 °C. Response and response time variation with different NiO doping percentages were calculated. The calculations took into account the combustion of acetylene as it approached its autoignition temperature, which was not considered in previous works. The results show that the optimum response operating temperature of the C2H2 gas sensor is below the autoignition temperature of acetylene at 300 °C. A good agreement of theoretical response and response time with variation of temperature and acetylene concentration was obtained with the experiment. This study was the first to take into account the autoignition of C2H2 in gas sensor calculations and noted that gases can be easily distinguished by their autoignition temperature.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Mudar A. Abdulsattar

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



