On the Bromination of Diethyl 3,5-dimethyl-1H-pyrrole-2,4-dicarboxylate: the True Structure of the Halbig’s Product
DOI:
https://doi.org/10.31489/2959-0663/1-24-4Keywords:
Knorr pyrrole, α-methylpyrroles, bromination, the Halbig’s product, bromo derivatives, Diethyl 3,5-dimethyl-1H-pyrrole-2,4-dicarboxylate, phosphorylation, reaction mechanismAbstract
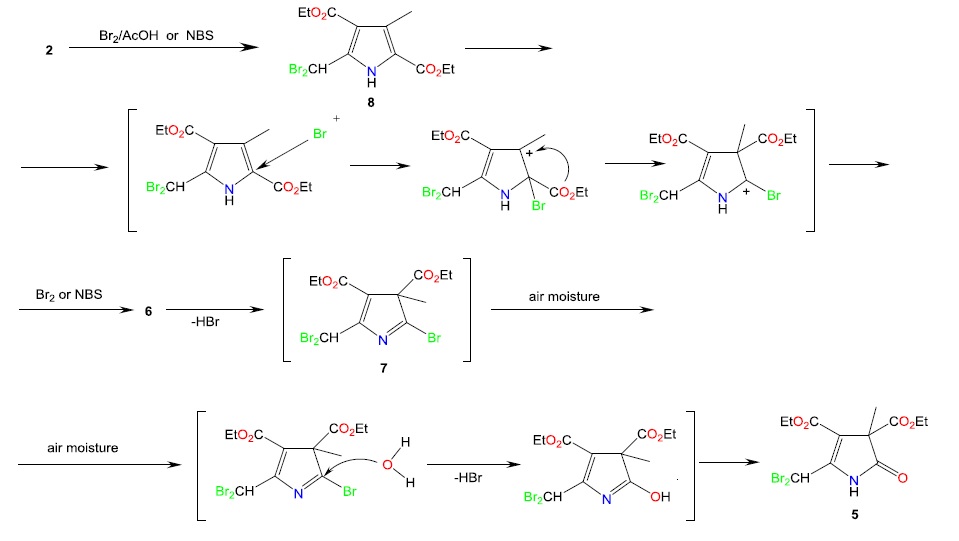
Pyrrole and its derivatives have attracted the attention of researchers as promising objects of practical importance in various areas of human life. Focusing on Knorr pyrrole as the most readily available pyrrole derivative, our objective was to obtain its bromo derivatives, containing an allyl bromine atom followed by phosphorylation reactions. The reaction involving bromination was carried out with 2 mol of N-bromosuccinimide per 1 mol of Knorr pyrrole 1 in carbon tetrachloride. This reaction was initiated with azobis(isobutyronitrile) and carried out at a boiling point. In acetic acid, the bromination reaction was carried out at a temperature of 38-45°C with a molar ratio of Knorr pyrrole to bromine of 1:4 and another reaction was carried out at 40-50°C with a molar ratio of Knorr pyrrole to bromine of 1:5. The bromination reaction was also performed in chloroform at 27-30°C in the presence of catalytic amounts of aluminum chloride. The same crystalline product was obtained in all cases. X-ray diffraction analysis revealed that the single crystal structure is diethyl 5-(dibromomethyl)-3-methyl-2-oxo-2,3-dihydro-1H-pyrrole-3,4-dicarboxylate 5 with two bromine atoms. This composition is identical to that of the Halbig product. It was found that the bromination of Knorr pyrrole 1 occurs stepwise with excess N-bromosuccinimide and bromine, resulting in the unsaturated lactam 5, commonly known as Halbig’s product. The true structure of product 5 was determined.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



