Quantifying Curcumin, Gallic Acid, and Resveratrol in a Polyherbal Mixture: a Robust HPTLC Method
DOI:
https://doi.org/10.31489/2959-0663/1-24-2Keywords:
HPTLC, curcumin, gallic acid, resveratrol, simultaneous estimation, standardization, anti-oxidant, validation, polyherbal mixtureAbstract
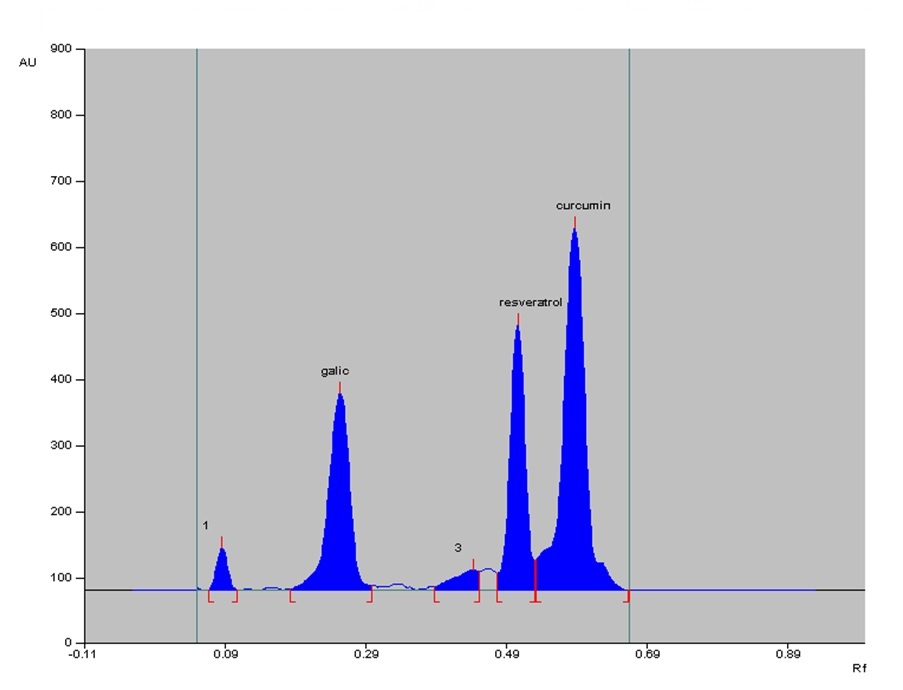
The need for this study arises from the growing importance of polyherbal formulations in herbal medicine, particularly due to their potential health benefits and synergistic effects. In this research, development and validation of an HPTLC method for simultaneous quantification of curcumin, gallic acid, and resveratrol in a polyherbal mixture are primary focus. Additionally, study assesses the Total Phenolic Content (TPC), Total Flavonoid Content (TFC) and antioxidant potential using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. The results reveal that TPC is 212.1 mg gallic acid equivalent per gram of plant material, and the TFC is 133.06 mg rutin equivalent per gram of plant material. Notably, DPPH assay demonstrates mixture's robust antioxidant activity, with an IC50 value of 3.83 µg/ml. Furthermore, HPTLC method used a carefully optimized mobile phase, providing excellent resolution for gallic acid, resveratrol, and curcumin with corresponding Rf values of 0.18, 0.43, and 0.53. This method is highly linear over a concentration range of 200-1000 ng/band for all three compounds. Precision studies confirm method's reproducibility with low Relative Standard Deviation values below 2 %. Ultimately, this HPTLC method offers a simple, accurate, precise, and cost-effective solution for routine quality control analysis, shedding light on potential applications of the polyherbal mixture in various therapeutic contexts.
Downloads
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.



